This project is not resposnive.
Mouse-over groups or click individual elements to get more information.
1
Element: Hydrogen
Series: Reactive Nonmetals
Atomic Number: 1
Energy Levels: 1
Atomic Weight: 1.008 u
Electronegativity: 2.20
Melting Point: 259.1° C
Boiling Point: -252.9° C
Electron Affinity: 72.8 kJ/mol
Ionization: 1,312.0 kJ/mol
Radius Calculated: 53 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Conductivity, Thermal: 0.1805 W/mK
Heat, Specific: 14,300 J/kgK
Abundance, Universe: 75%
Discovered: 1766
Series: Reactive Nonmetals
Atomic Number: 1
Energy Levels: 1
Atomic Weight: 1.008 u
Electronegativity: 2.20
Melting Point: 259.1° C
Boiling Point: -252.9° C
Electron Affinity: 72.8 kJ/mol
Ionization: 1,312.0 kJ/mol
Radius Calculated: 53 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Conductivity, Thermal: 0.1805 W/mK
Heat, Specific: 14,300 J/kgK
Abundance, Universe: 75%
Discovered: 1766
1
1
H
Hydrogen
1.008
Element: Helium
Series: Nobel Gas
Atomic Number: 2
Energy Levels: 2
Atomic Weight: 4.002602 u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: -269° C
Electron Affinity: 0 kJ/mol
Ionization: 2,372.3 kJ/mol
Radius Calculated: 31 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Conductivity, Thermal: 0.1785 W/mK
Heat, Specific: 5,193.1 J/kgK
Abundance, Universe: 23%
Discovered: 1895
Series: Nobel Gas
Atomic Number: 2
Energy Levels: 2
Atomic Weight: 4.002602 u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: -269° C
Electron Affinity: 0 kJ/mol
Ionization: 2,372.3 kJ/mol
Radius Calculated: 31 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Conductivity, Thermal: 0.1785 W/mK
Heat, Specific: 5,193.1 J/kgK
Abundance, Universe: 23%
Discovered: 1895
2
2
He
Helium
4.0026
2
Element: Lithium
Series: Alkali Metal
Atomic Number: 3
Energy Levels: 2, 1
Atomic Weight: 6.94 u
Electronegativity: 0.98
Melting Point: 180.54° C
Boiling Point: 1,342° C
Electron Affinity: 59.6 kJ/mol
Ionization, First: 520.2 kJ/mol
Radius Calculated: 167 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 11 GPa
Density, STP: 535 kg/m3 W/mK
Heat, Specific: 3,570 J/kgK
Abundance, Universe: 6.0E-7 % Discovered: 18172
Series: Alkali Metal
Atomic Number: 3
Energy Levels: 2, 1
Atomic Weight: 6.94 u
Electronegativity: 0.98
Melting Point: 180.54° C
Boiling Point: 1,342° C
Electron Affinity: 59.6 kJ/mol
Ionization, First: 520.2 kJ/mol
Radius Calculated: 167 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 11 GPa
Density, STP: 535 kg/m3 W/mK
Heat, Specific: 3,570 J/kgK
Abundance, Universe: 6.0E-7 % Discovered: 1817
3
2
1
Li
Lithium
6.94
Element: Berylium
Series: Alkaline Earth Metal
Atomic Number: 4
Energy Levels: 2, 2
Atomic Weight: 9.0121831 u
Electronegativity: 1.57
Melting Point: 1,287° C
Boiling Point: 2,470° C
Electron Affinity: 0 kJ/mol
Ionization, First: 899.5 kJ/mol
Radius Calculated: 112 pm
Hardness, Brinell: 600 MPa
Modulus, Bulk: 130 GPa
Density, STP: 1848 kg/m3 W/mK
Heat, Specific: 1,820 J/kgK
Conductivity, Thermal: 190 W/mK Abundance, Universe: 1.00E-7 % Discovered: 17972
Series: Alkaline Earth Metal
Atomic Number: 4
Energy Levels: 2, 2
Atomic Weight: 9.0121831 u
Electronegativity: 1.57
Melting Point: 1,287° C
Boiling Point: 2,470° C
Electron Affinity: 0 kJ/mol
Ionization, First: 899.5 kJ/mol
Radius Calculated: 112 pm
Hardness, Brinell: 600 MPa
Modulus, Bulk: 130 GPa
Density, STP: 1848 kg/m3 W/mK
Heat, Specific: 1,820 J/kgK
Conductivity, Thermal: 190 W/mK Abundance, Universe: 1.00E-7 % Discovered: 1797
4
2
2
Be
Berylium
9.0122
Element: Carbon
Series: Reactive Nonmetal
Atomic Number: 6
Energy Levels: 2, 4
Atomic Weight: 12.011 u
Electronegativity: 2.55
Melting Point: 3,642° C
Boiling Point: 3,642° C
Electron Affinity: 153.9 kJ/mol
Ionization, First: 1,086.5 kJ/mol
Radius Calculated: 67 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 33 GPa
Density, STP: 2,260 kg/m3 W/mK
Heat, Specific: 710 J/kgK
Conductivity, Thermal: 140 W/mK Abundance, Universe: 0.50 % Discovered: 3750 BCE2
Series: Reactive Nonmetal
Atomic Number: 6
Energy Levels: 2, 4
Atomic Weight: 12.011 u
Electronegativity: 2.55
Melting Point: 3,642° C
Boiling Point: 3,642° C
Electron Affinity: 153.9 kJ/mol
Ionization, First: 1,086.5 kJ/mol
Radius Calculated: 67 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 33 GPa
Density, STP: 2,260 kg/m3 W/mK
Heat, Specific: 710 J/kgK
Conductivity, Thermal: 140 W/mK Abundance, Universe: 0.50 % Discovered: 3750 BCE
6
2
4
C
Carbon
12.011
Element: Nitrogen
Series: Reactive Nonmetal
Atomic Number: 7
Energy Levels: 2, 5
Atomic Weight: 14.007 u
Electronegativity: 3.04
Melting Point: -210.1° C
Boiling Point: -195.8° C
Electron Affinity: 7 kJ/mol
Ionization, First: 1,402.3 kJ/mol
Radius Calculated: 56 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 1.251 kg/m3 W/mK
Heat, Specific: 1,040 J/kgK
Conductivity, Thermal: 0.02583 W/mK Abundance, Universe: 0.100 % Discovered: 17722
Series: Reactive Nonmetal
Atomic Number: 7
Energy Levels: 2, 5
Atomic Weight: 14.007 u
Electronegativity: 3.04
Melting Point: -210.1° C
Boiling Point: -195.8° C
Electron Affinity: 7 kJ/mol
Ionization, First: 1,402.3 kJ/mol
Radius Calculated: 56 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 1.251 kg/m3 W/mK
Heat, Specific: 1,040 J/kgK
Conductivity, Thermal: 0.02583 W/mK Abundance, Universe: 0.100 % Discovered: 1772
7
2
5
N
Nitrogen
14.007
Element: Oxygen
Series: Reactive Nonmetal
Atomic Number: 8
Energy Levels: 2, 6
Atomic Weight: 15.999 u
Electronegativity: 3.44
Melting Point: -218° C
Boiling Point: -183° C
Electron Affinity: 141 kJ/mol
Ionization, First: 1,313.9 kJ/mol
Radius Calculated: 48 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 1.429 kg/m3 W/mK
Heat, Specific: 919 J/kgK
Conductivity, Thermal: 0.02658 W/mK Abundance, Universe: 1.00 % Discovered: 17742
Series: Reactive Nonmetal
Atomic Number: 8
Energy Levels: 2, 6
Atomic Weight: 15.999 u
Electronegativity: 3.44
Melting Point: -218° C
Boiling Point: -183° C
Electron Affinity: 141 kJ/mol
Ionization, First: 1,313.9 kJ/mol
Radius Calculated: 48 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 1.429 kg/m3 W/mK
Heat, Specific: 919 J/kgK
Conductivity, Thermal: 0.02658 W/mK Abundance, Universe: 1.00 % Discovered: 1774
8
2
6
O
Oxygen
15.999
Element: Fluorine
Series: Reactive Nonmetal
Atomic Number: 9
Energy Levels: 2, 7
Atomic Weight: 18.998403163 u
Electronegativity: 3.98
Melting Point: -220° C
Boiling Point: -188.1° C
Electron Affinity: 328 kJ/mol
Ionization, First: 1,681.0 kJ/mol
Radius Calculated: 42 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 1.696 kg/m3 W/mK
Heat, Specific: 824 J/kgK
Conductivity, Thermal: 0.0277 W/mK Abundance, Universe: 0.000040 % Discovered: 18862
Series: Reactive Nonmetal
Atomic Number: 9
Energy Levels: 2, 7
Atomic Weight: 18.998403163 u
Electronegativity: 3.98
Melting Point: -220° C
Boiling Point: -188.1° C
Electron Affinity: 328 kJ/mol
Ionization, First: 1,681.0 kJ/mol
Radius Calculated: 42 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 1.696 kg/m3 W/mK
Heat, Specific: 824 J/kgK
Conductivity, Thermal: 0.0277 W/mK Abundance, Universe: 0.000040 % Discovered: 1886
9
2
7
F
Fluorine
18.998
Element: Neon
Series: Nobel Gas
Atomic Number: 10
Energy Levels: 2, 8
Atomic Weight: 20.1797 u
Electronegativity: N/A
Melting Point: -248.6° C
Boiling Point: -246.1° C
Electron Affinity: 0 kJ/mol
Ionization, First: 2,080.7 kJ/mol
Radius Calculated: 38 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 0.900 kg/m3 W/mK
Conductivity, Thermal: 0.0491 W/mK
Heat, Specific: 1,030.0 J/kgK
Abundance, Universe: 0.13 %
Discovered: 18982
Series: Nobel Gas
Atomic Number: 10
Energy Levels: 2, 8
Atomic Weight: 20.1797 u
Electronegativity: N/A
Melting Point: -248.6° C
Boiling Point: -246.1° C
Electron Affinity: 0 kJ/mol
Ionization, First: 2,080.7 kJ/mol
Radius Calculated: 38 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 0.900 kg/m3 W/mK
Conductivity, Thermal: 0.0491 W/mK
Heat, Specific: 1,030.0 J/kgK
Abundance, Universe: 0.13 %
Discovered: 1898
10
2
8
Ne
Neon
20.180
3
Element: Sodium
Series: Alkali Metals
Atomic Number: 11
Energy Levels: 2, 8, 1
Atomic Weight: 22.98976928 u
Electronegativity: 0.93
Melting Point: 97.720° C
Boiling Point: 882.9° C
Electron Affinity: 52.8 kJ/mol
Ionization, First: 495.8 kJ/mol
Radius Calculated: 190 pm
Hardness, Brinell: 0.69 MPa
Modulus, Bulk: 6.3 GPa
Density, STP: 968 kg/m3 W/mK
Conductivity, Thermal: 140 W/mK
Heat, Specific: 1,230.0 J/kgK
Abundance, Universe: 0.0020 %
Discovered: 18072
Series: Alkali Metals
Atomic Number: 11
Energy Levels: 2, 8, 1
Atomic Weight: 22.98976928 u
Electronegativity: 0.93
Melting Point: 97.720° C
Boiling Point: 882.9° C
Electron Affinity: 52.8 kJ/mol
Ionization, First: 495.8 kJ/mol
Radius Calculated: 190 pm
Hardness, Brinell: 0.69 MPa
Modulus, Bulk: 6.3 GPa
Density, STP: 968 kg/m3 W/mK
Conductivity, Thermal: 140 W/mK
Heat, Specific: 1,230.0 J/kgK
Abundance, Universe: 0.0020 %
Discovered: 1807
11
2
8
1
Na
Sodium
22.990
Element: Magnesium
Series: Alkaline Earth Metals
Atomic Number: 12
Energy Levels: 2, 8, 2
Atomic Weight: 24.305 u
Electronegativity: 1.31
Melting Point: 650° C
Boiling Point: 1,090° C
Electron Affinity: 0 kJ/mol
Ionization, First: 737.7 kJ/mol
Radius Calculated: 145 pm
Hardness, Brinell: 260 MPa
Modulus, Bulk: 45 GPa
Density, STP: 1,738 kg/m3 W/mK
Conductivity, Thermal: 160 W/mK
Heat, Specific: 1,020 J/kgK
Abundance, Universe: 0.060 %
Discovered: 17552
Series: Alkaline Earth Metals
Atomic Number: 12
Energy Levels: 2, 8, 2
Atomic Weight: 24.305 u
Electronegativity: 1.31
Melting Point: 650° C
Boiling Point: 1,090° C
Electron Affinity: 0 kJ/mol
Ionization, First: 737.7 kJ/mol
Radius Calculated: 145 pm
Hardness, Brinell: 260 MPa
Modulus, Bulk: 45 GPa
Density, STP: 1,738 kg/m3 W/mK
Conductivity, Thermal: 160 W/mK
Heat, Specific: 1,020 J/kgK
Abundance, Universe: 0.060 %
Discovered: 1755
12
2
8
2
Mg
Magnesium
24.305
Element: Aluminum
Series: Post Transition Metals
Atomic Number: 13
Energy Levels: 2, 8, 3
Atomic Weight: 26.9815385 u
Electronegativity: 1.61
Melting Point: 660.32° C
Boiling Point: 2,519° C
Electron Affinity: 42.5 kJ/mol
Ionization, First: 577.5 kJ/mol
Radius Calculated: 118 pm
Hardness, Brinell: 245 MPa
Modulus, Bulk: 76 GPa
Density, STP: 2,700 kg/m3 W/mK
Conductivity, Thermal: 235 W/mK
Heat, Specific: 904 J/kgK
Abundance, Universe: 0.0050 %
Discovered: 18252
Series: Post Transition Metals
Atomic Number: 13
Energy Levels: 2, 8, 3
Atomic Weight: 26.9815385 u
Electronegativity: 1.61
Melting Point: 660.32° C
Boiling Point: 2,519° C
Electron Affinity: 42.5 kJ/mol
Ionization, First: 577.5 kJ/mol
Radius Calculated: 118 pm
Hardness, Brinell: 245 MPa
Modulus, Bulk: 76 GPa
Density, STP: 2,700 kg/m3 W/mK
Conductivity, Thermal: 235 W/mK
Heat, Specific: 904 J/kgK
Abundance, Universe: 0.0050 %
Discovered: 1825
13
2
8
3
Al
Aluminum
26.982
Element: Phosphorus
Series: Reactive NonMetal
Atomic Number: 15
Energy Levels: 2, 8, 5
Atomic Weight: 30.973761998 u
Electronegativity: 2.19
Melting Point: 44.15° C
Boiling Point: 280.5° C
Electron Affinity: 72 kJ/mol
Ionization, First: 1,011.8 kJ/mol
Radius Calculated: 98 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 11 GPa
Density, STP: 1,823 kg/m3 W/mK
Conductivity, Thermal: 0.236 W/mK
Heat, Specific: 769.7 J/kgK
Abundance, Universe: 0.00070 %
Discovered: 16692
Series: Reactive NonMetal
Atomic Number: 15
Energy Levels: 2, 8, 5
Atomic Weight: 30.973761998 u
Electronegativity: 2.19
Melting Point: 44.15° C
Boiling Point: 280.5° C
Electron Affinity: 72 kJ/mol
Ionization, First: 1,011.8 kJ/mol
Radius Calculated: 98 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 11 GPa
Density, STP: 1,823 kg/m3 W/mK
Conductivity, Thermal: 0.236 W/mK
Heat, Specific: 769.7 J/kgK
Abundance, Universe: 0.00070 %
Discovered: 1669
15
2
8
5
P
Phosphorus
30.974
Element: Sulfur
Series: Reactive NonMetal
Atomic Number: 16
Energy Levels: 2, 8, 6
Atomic Weight: 32.06 u
Electronegativity: 2.58
Melting Point: 115.21° C
Boiling Point: 444.72° C
Electron Affinity: 200 kJ/mol
Ionization, First: 999.6 kJ/mol
Radius Calculated: 88 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 7.7 GPa
Density, STP: 1,960 kg/m3 W/mK
Conductivity, Thermal: 0.205 W/mK
Heat, Specific: 705 J/kgK
Abundance, Universe: 0.050 %
Discovered: 500 BCE2
Series: Reactive NonMetal
Atomic Number: 16
Energy Levels: 2, 8, 6
Atomic Weight: 32.06 u
Electronegativity: 2.58
Melting Point: 115.21° C
Boiling Point: 444.72° C
Electron Affinity: 200 kJ/mol
Ionization, First: 999.6 kJ/mol
Radius Calculated: 88 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 7.7 GPa
Density, STP: 1,960 kg/m3 W/mK
Conductivity, Thermal: 0.205 W/mK
Heat, Specific: 705 J/kgK
Abundance, Universe: 0.050 %
Discovered: 500 BCE
16
2
8
6
S
Sulfur
32.06
Element: Chlorine
Series: Reactive NonMetal
Atomic Number: 17
Energy Levels: 2, 8, 7
Atomic Weight: 35.45 u
Electronegativity: 3.16
Melting Point: -101.5° C
Boiling Point: -34.040° C
Electron Affinity: 349 kJ/mol
Ionization, First: 1.251.2 kJ/mol
Radius Calculated: 79 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 1.1 GPa
Density, STP: 3.214 kg/m3 W/mK
Conductivity, Thermal: 0.0089 W/mK
Heat, Specific: 478.2 J/kgK
Abundance, Universe: 0.000100 %
Discovered: 17742
Series: Reactive NonMetal
Atomic Number: 17
Energy Levels: 2, 8, 7
Atomic Weight: 35.45 u
Electronegativity: 3.16
Melting Point: -101.5° C
Boiling Point: -34.040° C
Electron Affinity: 349 kJ/mol
Ionization, First: 1.251.2 kJ/mol
Radius Calculated: 79 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 1.1 GPa
Density, STP: 3.214 kg/m3 W/mK
Conductivity, Thermal: 0.0089 W/mK
Heat, Specific: 478.2 J/kgK
Abundance, Universe: 0.000100 %
Discovered: 1774
17
2
8
7
Cl
Chlorine
35.45
Element: Argon
Series: Nobel Gas
Atomic Number: 18
Energy Levels: 2, 8, 8
Atomic Weight: 39.948 u
Electronegativity: N/A
Melting Point: -189° C
Boiling Point: -186° C
Electron Affinity: 0 kJ/mol
Ionization, First: 1,520.6 kJ/mol
Radius Calculated: 71 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 1.784 kg/m3 W/mK
Conductivity, Thermal: 0.01772 W/mK
Heat, Specific: 520.33 J/kgK
Abundance, Universe: 0.020 %
Discovered: 18942
Series: Nobel Gas
Atomic Number: 18
Energy Levels: 2, 8, 8
Atomic Weight: 39.948 u
Electronegativity: N/A
Melting Point: -189° C
Boiling Point: -186° C
Electron Affinity: 0 kJ/mol
Ionization, First: 1,520.6 kJ/mol
Radius Calculated: 71 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 1.784 kg/m3 W/mK
Conductivity, Thermal: 0.01772 W/mK
Heat, Specific: 520.33 J/kgK
Abundance, Universe: 0.020 %
Discovered: 1894
18
2
8
8
Ar
Argon
39.948
4
Element: Potassium
Series: Alkali Metal
Atomic Number: 19
Energy Levels: 2, 8, 8, 1
Atomic Weight: 39.0983 u
Electronegativity: 0.82
Melting Point: 63.380° C
Boiling Point: 758.9° C
Electron Affinity: 48.4 kJ/mol
Ionization, First: 418.8 kJ/mol
Radius Calculated: 243 pm
Hardness, Brinell: 0.363 MPa
Modulus, Bulk: 3.1 GPa
Density, STP: 856 kg/m3 W/mK
Conductivity, Thermal: 100 W/mK
Heat, Specific: 757 J/kgK
Abundance, Universe: 0.00030 %
Discovered: 18072
Series: Alkali Metal
Atomic Number: 19
Energy Levels: 2, 8, 8, 1
Atomic Weight: 39.0983 u
Electronegativity: 0.82
Melting Point: 63.380° C
Boiling Point: 758.9° C
Electron Affinity: 48.4 kJ/mol
Ionization, First: 418.8 kJ/mol
Radius Calculated: 243 pm
Hardness, Brinell: 0.363 MPa
Modulus, Bulk: 3.1 GPa
Density, STP: 856 kg/m3 W/mK
Conductivity, Thermal: 100 W/mK
Heat, Specific: 757 J/kgK
Abundance, Universe: 0.00030 %
Discovered: 1807
19
2
8
8
1
K
Potassium
39.098
Element: Calcium
Series: Alkaline Earth Metal
Atomic Number: 20
Energy Levels: 2, 8, 8, 2
Atomic Weight: 40.078 u
Electronegativity: 1.0
Melting Point: 841.9° C
Boiling Point: 1,484° C
Electron Affinity: 2.37 kJ/mol
Ionization, First: 589.8 kJ/mol
Radius Calculated: 194 pm
Hardness, Brinell: 167 MPa
Modulus, Bulk: 17 GPa
Density, STP: 1,550 kg/m3 W/mK
Conductivity, Thermal: 200 W/mK
Heat, Specific: 631 J/kgK
Abundance, Universe: 0.0070 %
Discovered: 18082
Series: Alkaline Earth Metal
Atomic Number: 20
Energy Levels: 2, 8, 8, 2
Atomic Weight: 40.078 u
Electronegativity: 1.0
Melting Point: 841.9° C
Boiling Point: 1,484° C
Electron Affinity: 2.37 kJ/mol
Ionization, First: 589.8 kJ/mol
Radius Calculated: 194 pm
Hardness, Brinell: 167 MPa
Modulus, Bulk: 17 GPa
Density, STP: 1,550 kg/m3 W/mK
Conductivity, Thermal: 200 W/mK
Heat, Specific: 631 J/kgK
Abundance, Universe: 0.0070 %
Discovered: 1808
20
2
8
8
2
Ca
Calcium
40.078
Element: Scandium
Series: Transition Metal
Atomic Number: 21
Energy Levels: 2, 8, 9, 2
Atomic Weight: 44.955908 u
Electronegativity: 1.36
Melting Point: 1,541° C
Boiling Point: 2,830° C
Electron Affinity: 18.1 kJ/mol
Ionization, First: 633.1 kJ/mol
Radius Calculated: 184 pm
Hardness, Brinell: 750 MPa
Modulus, Bulk: 57 GPa
Density, STP: 2,985 kg/m3 W/mK
Conductivity, Thermal: 16 W/mK
Heat, Specific: 567 J/kgK
Abundance, Universe: 3.0E-6 %
Discovered: 18792
Series: Transition Metal
Atomic Number: 21
Energy Levels: 2, 8, 9, 2
Atomic Weight: 44.955908 u
Electronegativity: 1.36
Melting Point: 1,541° C
Boiling Point: 2,830° C
Electron Affinity: 18.1 kJ/mol
Ionization, First: 633.1 kJ/mol
Radius Calculated: 184 pm
Hardness, Brinell: 750 MPa
Modulus, Bulk: 57 GPa
Density, STP: 2,985 kg/m3 W/mK
Conductivity, Thermal: 16 W/mK
Heat, Specific: 567 J/kgK
Abundance, Universe: 3.0E-6 %
Discovered: 1879
21
2
8
9
2
Sc
Scandium
44.956
Element: Titanium
Series: Transition Metal
Atomic Number: 22
Energy Levels: 2, 8, 10, 2
Atomic Weight: 47.867 u
Electronegativity: 1.54
Melting Point: 1,668° C
Boiling Point: 3,287° C
Electron Affinity: 7.6 kJ/mol
Ionization, First: 658.8 kJ/mol
Radius Calculated: 176 pm
Hardness, Brinell: 716 MPa
Modulus, Bulk: 110 GPa
Density, STP: 4,507 kg/m3 W/mK
Conductivity, Thermal: 22 W/mK
Heat, Specific: 520 J/kgK
Abundance, Universe: 0.00030 %
Discovered: 17912
Series: Transition Metal
Atomic Number: 22
Energy Levels: 2, 8, 10, 2
Atomic Weight: 47.867 u
Electronegativity: 1.54
Melting Point: 1,668° C
Boiling Point: 3,287° C
Electron Affinity: 7.6 kJ/mol
Ionization, First: 658.8 kJ/mol
Radius Calculated: 176 pm
Hardness, Brinell: 716 MPa
Modulus, Bulk: 110 GPa
Density, STP: 4,507 kg/m3 W/mK
Conductivity, Thermal: 22 W/mK
Heat, Specific: 520 J/kgK
Abundance, Universe: 0.00030 %
Discovered: 1791
22
2
8
10
2
Ti
Titanium
47.867
Element: Vanadium
Series: Transition Metal
Atomic Number: 23
Energy Levels: 2, 8, 11, 2
Atomic Weight: 50.9415 u
Electronegativity: 1.63
Melting Point: 1,910° C
Boiling Point: 3,407° C
Electron Affinity: 50.6 kJ/mol
Ionization, First: 650.9 kJ/mol
Radius Calculated: 171 pm
Hardness, Brinell: 628 MPa
Modulus, Bulk: 160 GPa
Density, STP: 6,110 kg/m3 W/mK
Conductivity, Thermal: 31 W/mK
Heat, Specific: 489 J/kgK
Abundance, Universe: 0.000100 %
Discovered: 18012
Series: Transition Metal
Atomic Number: 23
Energy Levels: 2, 8, 11, 2
Atomic Weight: 50.9415 u
Electronegativity: 1.63
Melting Point: 1,910° C
Boiling Point: 3,407° C
Electron Affinity: 50.6 kJ/mol
Ionization, First: 650.9 kJ/mol
Radius Calculated: 171 pm
Hardness, Brinell: 628 MPa
Modulus, Bulk: 160 GPa
Density, STP: 6,110 kg/m3 W/mK
Conductivity, Thermal: 31 W/mK
Heat, Specific: 489 J/kgK
Abundance, Universe: 0.000100 %
Discovered: 1801
23
2
8
11
2
V
Vanadium
50.942
Element: Chromium
Series: Transition Metal
Atomic Number: 24
Energy Levels: 2, 8, 13, 1
Atomic Weight: 51.9961 u
Electronegativity: 1.66
Melting Point: 1,907° C
Boiling Point: 2,671° C
Electron Affinity: 64.3 kJ/mol
Ionization, First: 652.9 kJ/mol
Radius Calculated: 166 pm
Hardness, Brinell: 1,120 MPa
Modulus, Bulk: 160 GPa
Density, STP: 7,190 kg/m3 W/mK
Conductivity, Thermal: 94 W/mK
Heat, Specific: 448 J/kgK
Abundance, Universe: 0.0015 %
Discovered: 17972
Series: Transition Metal
Atomic Number: 24
Energy Levels: 2, 8, 13, 1
Atomic Weight: 51.9961 u
Electronegativity: 1.66
Melting Point: 1,907° C
Boiling Point: 2,671° C
Electron Affinity: 64.3 kJ/mol
Ionization, First: 652.9 kJ/mol
Radius Calculated: 166 pm
Hardness, Brinell: 1,120 MPa
Modulus, Bulk: 160 GPa
Density, STP: 7,190 kg/m3 W/mK
Conductivity, Thermal: 94 W/mK
Heat, Specific: 448 J/kgK
Abundance, Universe: 0.0015 %
Discovered: 1797
24
2
8
13
1
Cr
Chromium
51.996
Element: Manganese
Series: Transition Metal
Atomic Number: 25
Energy Levels: 2, 8, 13, 2
Atomic Weight: 54.938044 u
Electronegativity: 1.55
Melting Point: 1,246° C
Boiling Point: 2,061° C
Electron Affinity: 0 kJ/mol
Ionization, First: 717.3 kJ/mol
Radius Calculated: 161 pm
Hardness, Brinell: 196 MPa
Modulus, Bulk: 120 GPa
Density, STP: 7,470 kg/m3 W/mK
Conductivity, Thermal: 7.8 W/mK
Heat, Specific: 479 J/kgK
Abundance, Universe: 0.00080 %
Discovered: 17742
Series: Transition Metal
Atomic Number: 25
Energy Levels: 2, 8, 13, 2
Atomic Weight: 54.938044 u
Electronegativity: 1.55
Melting Point: 1,246° C
Boiling Point: 2,061° C
Electron Affinity: 0 kJ/mol
Ionization, First: 717.3 kJ/mol
Radius Calculated: 161 pm
Hardness, Brinell: 196 MPa
Modulus, Bulk: 120 GPa
Density, STP: 7,470 kg/m3 W/mK
Conductivity, Thermal: 7.8 W/mK
Heat, Specific: 479 J/kgK
Abundance, Universe: 0.00080 %
Discovered: 1774
25
2
8
13
2
Mn
Manganese
54.938
Element: Iron
Series: Transition Metal
Atomic Number: 26
Energy Levels: 2, 8, 14, 2
Atomic Weight: 55.845 u
Electronegativity: 1.83
Melting Point: 1,538° C
Boiling Point: 2,861° C
Electron Affinity: 15.7 kJ/mol
Ionization, First: 762.5 kJ/mol
Radius Calculated: 156 pm
Hardness, Brinell: 490 MPa
Modulus, Bulk: 170 GPa
Density, STP: 7,874 kg/m3 W/mK
Conductivity, Thermal: 80 W/mK
Heat, Specific: 449 J/kgK
Abundance, Universe: 0.11 %
Discovered: 2000 BCE2
Series: Transition Metal
Atomic Number: 26
Energy Levels: 2, 8, 14, 2
Atomic Weight: 55.845 u
Electronegativity: 1.83
Melting Point: 1,538° C
Boiling Point: 2,861° C
Electron Affinity: 15.7 kJ/mol
Ionization, First: 762.5 kJ/mol
Radius Calculated: 156 pm
Hardness, Brinell: 490 MPa
Modulus, Bulk: 170 GPa
Density, STP: 7,874 kg/m3 W/mK
Conductivity, Thermal: 80 W/mK
Heat, Specific: 449 J/kgK
Abundance, Universe: 0.11 %
Discovered: 2000 BCE
26
2
8
14
2
Fe
Iron
55.845
Element: Cobalt
Series: Transition Metal
Atomic Number: 27
Energy Levels: 2, 8, 15, 2
Atomic Weight: 58.933194 u
Electronegativity: 1.88
Melting Point: 1,495° C
Boiling Point: 2,900° C
Electron Affinity: 63.7 kJ/mol
Ionization, First: 760.4 kJ/mol
Radius Calculated: 152 pm
Hardness, Brinell: 700 MPa
Modulus, Bulk: 180 GPa
Density, STP: 8,900 kg/m3 W/mK
Conductivity, Thermal: 100 W/mK
Heat, Specific: 421 J/kgK
Abundance, Universe: 0.00030 %
Discovered: 17352
Series: Transition Metal
Atomic Number: 27
Energy Levels: 2, 8, 15, 2
Atomic Weight: 58.933194 u
Electronegativity: 1.88
Melting Point: 1,495° C
Boiling Point: 2,900° C
Electron Affinity: 63.7 kJ/mol
Ionization, First: 760.4 kJ/mol
Radius Calculated: 152 pm
Hardness, Brinell: 700 MPa
Modulus, Bulk: 180 GPa
Density, STP: 8,900 kg/m3 W/mK
Conductivity, Thermal: 100 W/mK
Heat, Specific: 421 J/kgK
Abundance, Universe: 0.00030 %
Discovered: 1735
27
2
8
15
2
Co
Cobalt
58.933
Element: Nickel
Series: Transition Metal
Atomic Number: 28
Energy Levels: 2, 8, 16, 2
Atomic Weight: 58.6934 u
Electronegativity: 1.91
Melting Point: 1,455° C
Boiling Point: 2,913° C
Electron Affinity: 112 kJ/mol
Ionization, First: 737.1 kJ/mol
Radius Calculated: 149 pm
Hardness, Brinell: 700 MPa
Modulus, Bulk: 180 GPa
Density, STP: 8,908 kg/m3 W/mK
Conductivity, Thermal: 91 W/mK
Heat, Specific: 445 J/kgK
Abundance, Universe: 0.0060 %
Discovered: 17512
Series: Transition Metal
Atomic Number: 28
Energy Levels: 2, 8, 16, 2
Atomic Weight: 58.6934 u
Electronegativity: 1.91
Melting Point: 1,455° C
Boiling Point: 2,913° C
Electron Affinity: 112 kJ/mol
Ionization, First: 737.1 kJ/mol
Radius Calculated: 149 pm
Hardness, Brinell: 700 MPa
Modulus, Bulk: 180 GPa
Density, STP: 8,908 kg/m3 W/mK
Conductivity, Thermal: 91 W/mK
Heat, Specific: 445 J/kgK
Abundance, Universe: 0.0060 %
Discovered: 1751
28
2
8
16
2
Ni
Nickel
58.693
Element: Copper
Series: Transition Metal
Atomic Number: 29
Energy Levels: 2, 8, 18, 1
Atomic Weight: 63.546 u
Electronegativity: 1.90
Melting Point: 1.084.62° C
Boiling Point: 2.562° C
Electron Affinity: 118.4 kJ/mol
Ionization, First: 745.5 kJ/mol
Radius Calculated: 145 pm
Hardness, Brinell: 874 MPa
Modulus, Bulk: 140 GPa
Density, STP: 8,960 kg/m3 W/mK
Conductivity, Thermal: 400 W/mK
Heat, Specific: 384.4 J/kgK
Abundance, Universe: 6.0E-6 %
Discovered: 8000 BCE2
Series: Transition Metal
Atomic Number: 29
Energy Levels: 2, 8, 18, 1
Atomic Weight: 63.546 u
Electronegativity: 1.90
Melting Point: 1.084.62° C
Boiling Point: 2.562° C
Electron Affinity: 118.4 kJ/mol
Ionization, First: 745.5 kJ/mol
Radius Calculated: 145 pm
Hardness, Brinell: 874 MPa
Modulus, Bulk: 140 GPa
Density, STP: 8,960 kg/m3 W/mK
Conductivity, Thermal: 400 W/mK
Heat, Specific: 384.4 J/kgK
Abundance, Universe: 6.0E-6 %
Discovered: 8000 BCE
29
2
8
18
1
Cu
Copper
63.546
Element: Zinc
Series: Transition Metal
Atomic Number: 29
Energy Levels: 2, 8, 18, 2
Atomic Weight: 65.38 u
Electronegativity: 1.65
Melting Point: 419.53° C
Boiling Point: 906.9° C
Electron Affinity: 0 kJ/mol
Ionization, First: 906.4 kJ/mol
Radius Calculated: 142 pm
Hardness, Brinell: 412 MPa
Modulus, Bulk: 70 GPa
Density, STP: 7,140 kg/m3 W/mK
Conductivity, Thermal: 120 W/mK
Heat, Specific: 388 J/kgK
Abundance, Universe: 0.000030 %
Discovered: 15002
Series: Transition Metal
Atomic Number: 29
Energy Levels: 2, 8, 18, 2
Atomic Weight: 65.38 u
Electronegativity: 1.65
Melting Point: 419.53° C
Boiling Point: 906.9° C
Electron Affinity: 0 kJ/mol
Ionization, First: 906.4 kJ/mol
Radius Calculated: 142 pm
Hardness, Brinell: 412 MPa
Modulus, Bulk: 70 GPa
Density, STP: 7,140 kg/m3 W/mK
Conductivity, Thermal: 120 W/mK
Heat, Specific: 388 J/kgK
Abundance, Universe: 0.000030 %
Discovered: 1500
30
2
8
18
2
Zn
Zinc
65.38
Element: Gallium
Series: Post Transition Metal
Atomic Number: 30
Energy Levels: 2, 8, 18, 3
Atomic Weight: 69.723 u
Electronegativity: 1.81
Melting Point: 29.760° C
Boiling Point: 2,204° C
Electron Affinity: 28.9 kJ/mol
Ionization, First: 578.8 kJ/mol
Radius Calculated: 136 pm
Hardness, Brinell: 60 MPa
Modulus, Bulk: N/A GPa
Density, STP: 5,904 kg/m3 W/mK
Conductivity, Thermal: 29 W/mK
Heat, Specific: 371 J/kgK
Abundance, Universe: 1.00E-6 %
Discovered: 18752
Series: Post Transition Metal
Atomic Number: 30
Energy Levels: 2, 8, 18, 3
Atomic Weight: 69.723 u
Electronegativity: 1.81
Melting Point: 29.760° C
Boiling Point: 2,204° C
Electron Affinity: 28.9 kJ/mol
Ionization, First: 578.8 kJ/mol
Radius Calculated: 136 pm
Hardness, Brinell: 60 MPa
Modulus, Bulk: N/A GPa
Density, STP: 5,904 kg/m3 W/mK
Conductivity, Thermal: 29 W/mK
Heat, Specific: 371 J/kgK
Abundance, Universe: 1.00E-6 %
Discovered: 1875
31
2
8
18
3
Ga
Gallium
69.723
Element: Selenium
Series: Reactive NonMetal
Atomic Number: 34
Energy Levels: 2, 8, 18, 6
Atomic Weight: 78.971 u
Electronegativity: 2.55
Melting Point: 221° C
Boiling Point: 685° C
Electron Affinity: 195 kJ/mol
Ionization, First: 9410.0 kJ/mol
Radius Calculated: 103 pm
Hardness, Brinell: 736 MPa
Modulus, Bulk: 8.3 GPa
Density, STP: 4,819 kg/m3 W/mK
Conductivity, Thermal: 0.52 W/mK
Heat, Specific: 312.2 J/kgK
Abundance, Universe: 3.0E-6 %
Discovered: 18172
Series: Reactive NonMetal
Atomic Number: 34
Energy Levels: 2, 8, 18, 6
Atomic Weight: 78.971 u
Electronegativity: 2.55
Melting Point: 221° C
Boiling Point: 685° C
Electron Affinity: 195 kJ/mol
Ionization, First: 9410.0 kJ/mol
Radius Calculated: 103 pm
Hardness, Brinell: 736 MPa
Modulus, Bulk: 8.3 GPa
Density, STP: 4,819 kg/m3 W/mK
Conductivity, Thermal: 0.52 W/mK
Heat, Specific: 312.2 J/kgK
Abundance, Universe: 3.0E-6 %
Discovered: 1817
34
2
8
18
6
Se
Selenium
78.971
Element: Bromine
Series: Reactive NonMetal
Atomic Number: 35
Energy Levels: 2, 8, 18, 7
Atomic Weight: 79.904 u
Electronegativity: 2.96
Melting Point: -7.350° C
Boiling Point: 58.9° C
Electron Affinity: 324.6 kJ/mol
Ionization, First: 1,139.9 kJ/mol
Radius Calculated: 94 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 1.9 GPa
Density, STP: 3,120 kg/m3 W/mK
Conductivity, Thermal: 0.12 W/mK
Heat, Specific: 947.3 J/kgK
Abundance, Universe: 7.0E-7 %
Discovered: 18262
Series: Reactive NonMetal
Atomic Number: 35
Energy Levels: 2, 8, 18, 7
Atomic Weight: 79.904 u
Electronegativity: 2.96
Melting Point: -7.350° C
Boiling Point: 58.9° C
Electron Affinity: 324.6 kJ/mol
Ionization, First: 1,139.9 kJ/mol
Radius Calculated: 94 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 1.9 GPa
Density, STP: 3,120 kg/m3 W/mK
Conductivity, Thermal: 0.12 W/mK
Heat, Specific: 947.3 J/kgK
Abundance, Universe: 7.0E-7 %
Discovered: 1826
35
2
8
18
7
Br
Bromine
79.904
Element: Krypton
Series: Nobel Gas
Atomic Number: 36
Energy Levels: 2, 8, 18, 8
Atomic Weight: 83.798 u
Electronegativity: 3.0
Melting Point: -157.36° C
Boiling Point: -153.22° C
Electron Affinity: 0 kJ/mol
Ionization, First: 1.350.8 kJ/mol
Radius Calculated: 88 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 3.75 kg/m3 W/mK
Conductivity, Thermal: 0.00943 W/mK
Heat, Specific: 248.05 J/kgK
Abundance, Universe: 4.0E-6 %
Discovered: 18982
Series: Nobel Gas
Atomic Number: 36
Energy Levels: 2, 8, 18, 8
Atomic Weight: 83.798 u
Electronegativity: 3.0
Melting Point: -157.36° C
Boiling Point: -153.22° C
Electron Affinity: 0 kJ/mol
Ionization, First: 1.350.8 kJ/mol
Radius Calculated: 88 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 3.75 kg/m3 W/mK
Conductivity, Thermal: 0.00943 W/mK
Heat, Specific: 248.05 J/kgK
Abundance, Universe: 4.0E-6 %
Discovered: 1898
36
2
8
18
8
Kr
Krypton
83.798
5
Element: Rubidium
Series: Alkali Metal
Atomic Number: 37
Energy Levels: 2, 8, 18, 8, 1
Atomic Weight: 85.4678 u
Electronegativity: 0.82
Melting Point: 39.310° C
Boiling Point: 688° C
Electron Affinity: 46.9 kJ/mol
Ionization, First: 403.0 kJ/mol
Radius Calculated: 265 pm
Hardness, Brinell: 0.216 MPa
Modulus, Bulk: 2.5 GPa
Density, STP: 1,532 kg/m3 W/mK
Conductivity, Thermal: 58 W/mK
Heat, Specific: 364 J/kgK
Abundance, Universe: 1.00E-6 %
Discovered: 18612
Series: Alkali Metal
Atomic Number: 37
Energy Levels: 2, 8, 18, 8, 1
Atomic Weight: 85.4678 u
Electronegativity: 0.82
Melting Point: 39.310° C
Boiling Point: 688° C
Electron Affinity: 46.9 kJ/mol
Ionization, First: 403.0 kJ/mol
Radius Calculated: 265 pm
Hardness, Brinell: 0.216 MPa
Modulus, Bulk: 2.5 GPa
Density, STP: 1,532 kg/m3 W/mK
Conductivity, Thermal: 58 W/mK
Heat, Specific: 364 J/kgK
Abundance, Universe: 1.00E-6 %
Discovered: 1861
37
2
8
18
8
1
Rb
Rubidium
85.468
Element: Strontium
Series: Alkaline Earth Metal
Atomic Number: 38
Energy Levels: 2, 8, 18, 8, 2
Atomic Weight: 87.62 u
Electronegativity: 0.95
Melting Point: 776.9° C
Boiling Point: 1,382° C
Electron Affinity: 5.03 kJ/mol
Ionization, First: 549.5 kJ/mol
Radius Calculated: 219 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 2,630 kg/m3 W/mK
Conductivity, Thermal: 35 W/mK
Heat, Specific: 300 J/kgK
Abundance, Universe: 4.0E-6 %
Discovered: 17902
Series: Alkaline Earth Metal
Atomic Number: 38
Energy Levels: 2, 8, 18, 8, 2
Atomic Weight: 87.62 u
Electronegativity: 0.95
Melting Point: 776.9° C
Boiling Point: 1,382° C
Electron Affinity: 5.03 kJ/mol
Ionization, First: 549.5 kJ/mol
Radius Calculated: 219 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 2,630 kg/m3 W/mK
Conductivity, Thermal: 35 W/mK
Heat, Specific: 300 J/kgK
Abundance, Universe: 4.0E-6 %
Discovered: 1790
38
2
8
18
8
2
Sr
Strontium
87.62
Element: Yttrium
Series: Transistion Metal
Atomic Number: 39
Energy Levels: 2, 8, 18, 9, 2
Atomic Weight: 88.90584 u
Electronegativity: 1.22
Melting Point: 1,526° C
Boiling Point: 3,345° C
Electron Affinity: 29.6 kJ/mol
Ionization, First: 600 kJ/mol
Radius Calculated: 212 pm
Hardness, Brinell: 589 MPa
Modulus, Bulk: 41 GPa
Density, STP: 4,472 kg/m3 W/mK
Conductivity, Thermal: 17 W/mK
Heat, Specific: 298 J/kgK
Abundance, Universe: 7.0E-7 %
Discovered: 17942
Series: Transistion Metal
Atomic Number: 39
Energy Levels: 2, 8, 18, 9, 2
Atomic Weight: 88.90584 u
Electronegativity: 1.22
Melting Point: 1,526° C
Boiling Point: 3,345° C
Electron Affinity: 29.6 kJ/mol
Ionization, First: 600 kJ/mol
Radius Calculated: 212 pm
Hardness, Brinell: 589 MPa
Modulus, Bulk: 41 GPa
Density, STP: 4,472 kg/m3 W/mK
Conductivity, Thermal: 17 W/mK
Heat, Specific: 298 J/kgK
Abundance, Universe: 7.0E-7 %
Discovered: 1794
39
2
8
18
9
2
Y
Yttrium
88.906
Element: Zirconium
Series: Transistion Metal
Atomic Number: 40
Energy Levels: 2, 8, 18, 10, 2
Atomic Weight: 91.224 u
Electronegativity: 1.33
Melting Point: 1,855° C
Boiling Point: 4,409° C
Electron Affinity: 41.1 kJ/mol
Ionization, First: 640.1 kJ/mol
Radius Calculated: 206 pm
Hardness, Brinell: 650 MPa
Modulus, Bulk: N/A GPa
Density, STP: 6,511 kg/m3 W/mK
Conductivity, Thermal: 23 W/mK
Heat, Specific: 278 J/kgK
Abundance, Universe: 5.0E-6 %
Discovered: 17892
Series: Transistion Metal
Atomic Number: 40
Energy Levels: 2, 8, 18, 10, 2
Atomic Weight: 91.224 u
Electronegativity: 1.33
Melting Point: 1,855° C
Boiling Point: 4,409° C
Electron Affinity: 41.1 kJ/mol
Ionization, First: 640.1 kJ/mol
Radius Calculated: 206 pm
Hardness, Brinell: 650 MPa
Modulus, Bulk: N/A GPa
Density, STP: 6,511 kg/m3 W/mK
Conductivity, Thermal: 23 W/mK
Heat, Specific: 278 J/kgK
Abundance, Universe: 5.0E-6 %
Discovered: 1789
40
2
8
18
10
2
Zr
Zirconium
91.224
Element: Niobium
Series: Transistion Metal
Atomic Number: 41
Energy Levels: 2, 8, 18, 12, 1
Atomic Weight: 92.90637 u
Electronegativity: 1.6
Melting Point: 2,477° C
Boiling Point: 4,744° C
Electron Affinity: 86.1 kJ/mol
Ionization, First: 652.1 kJ/mol
Radius Calculated: 198 pm
Hardness, Brinell: 736 MPa
Modulus, Bulk: 170 GPa
Density, STP: 8,570 kg/m3 W/mK
Conductivity, Thermal: 54 W/mK
Heat, Specific: 265 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 18012
Series: Transistion Metal
Atomic Number: 41
Energy Levels: 2, 8, 18, 12, 1
Atomic Weight: 92.90637 u
Electronegativity: 1.6
Melting Point: 2,477° C
Boiling Point: 4,744° C
Electron Affinity: 86.1 kJ/mol
Ionization, First: 652.1 kJ/mol
Radius Calculated: 198 pm
Hardness, Brinell: 736 MPa
Modulus, Bulk: 170 GPa
Density, STP: 8,570 kg/m3 W/mK
Conductivity, Thermal: 54 W/mK
Heat, Specific: 265 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 1801
41
2
8
18
12
1
Nb
Niobium
92.906
Element: Molybdenum
Series: Transition Metal
Atomic Number: 42
Energy Levels: 2, 8, 18, 13, 1
Atomic Weight: 95.95 u
Electronegativity: 2.16
Melting Point: 2,623° C
Boiling Point: 4,639° C
Electron Affinity: 71.9 kJ/mol
Ionization, First: 684.3 kJ/mol
Radius Calculated: 190 pm
Hardness, Brinell: 1,500 MPa
Modulus, Bulk: 230 GPa
Density, STP: 10,280 kg/m3 W/mK
Conductivity, Thermal: 139 W/mK
Heat, Specific: 251 J/kgK
Abundance, Universe: 5.0E-7 %
Discovered: 17812
Series: Transition Metal
Atomic Number: 42
Energy Levels: 2, 8, 18, 13, 1
Atomic Weight: 95.95 u
Electronegativity: 2.16
Melting Point: 2,623° C
Boiling Point: 4,639° C
Electron Affinity: 71.9 kJ/mol
Ionization, First: 684.3 kJ/mol
Radius Calculated: 190 pm
Hardness, Brinell: 1,500 MPa
Modulus, Bulk: 230 GPa
Density, STP: 10,280 kg/m3 W/mK
Conductivity, Thermal: 139 W/mK
Heat, Specific: 251 J/kgK
Abundance, Universe: 5.0E-7 %
Discovered: 1781
42
2
8
18
13
1
Mo
Molybdenum
95.95
Element: Technetium
Series: Transition Metal
Atomic Number: 43
Energy Levels: 2, 8, 18, 13, 2
Atomic Weight: (98) u
Electronegativity: 1.9
Melting Point: 2,157° C
Boiling Point: 4,265° C
Electron Affinity: 53 kJ/mol
Ionization, First: 702 kJ/mol
Radius Calculated: 183 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 11,500 kg/m3 W/mK
Conductivity, Thermal: 51 W/mK
Heat, Specific: 63 J/kgK
Abundance, Universe: 0 %
Discovered: 19372
Series: Transition Metal
Atomic Number: 43
Energy Levels: 2, 8, 18, 13, 2
Atomic Weight: (98) u
Electronegativity: 1.9
Melting Point: 2,157° C
Boiling Point: 4,265° C
Electron Affinity: 53 kJ/mol
Ionization, First: 702 kJ/mol
Radius Calculated: 183 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 11,500 kg/m3 W/mK
Conductivity, Thermal: 51 W/mK
Heat, Specific: 63 J/kgK
Abundance, Universe: 0 %
Discovered: 1937
43
2
8
18
13
2
Tc
Technetium
(98)
Element: Ruthenium
Series: Transition Metal
Atomic Number: 44
Energy Levels: 2, 8, 18, 15, 1
Atomic Weight: 101.07 u
Electronegativity: 2.2
Melting Point: 2,334° C
Boiling Point: 4,150° C
Electron Affinity: 101.3 kJ/mol
Ionization, First: 710.2 kJ/mol
Radius Calculated: 178 pm
Hardness, Brinell: 2,160 MPa
Modulus, Bulk: 220 GPa
Density, STP: 12,370 kg/m3 W/mK
Conductivity, Thermal: 120 W/mK
Heat, Specific: 238 J/kgK
Abundance, Universe: 4.0E-7 %
Discovered: 18442
Series: Transition Metal
Atomic Number: 44
Energy Levels: 2, 8, 18, 15, 1
Atomic Weight: 101.07 u
Electronegativity: 2.2
Melting Point: 2,334° C
Boiling Point: 4,150° C
Electron Affinity: 101.3 kJ/mol
Ionization, First: 710.2 kJ/mol
Radius Calculated: 178 pm
Hardness, Brinell: 2,160 MPa
Modulus, Bulk: 220 GPa
Density, STP: 12,370 kg/m3 W/mK
Conductivity, Thermal: 120 W/mK
Heat, Specific: 238 J/kgK
Abundance, Universe: 4.0E-7 %
Discovered: 1844
44
2
8
18
15
1
Ru
Ruthenium
101.07
Element: Rhodium
Series: Transition Metal
Atomic Number: 45
Energy Levels: 2, 8, 18, 16, 1
Atomic Weight: 102.90550 u
Electronegativity: 2.28
Melting Point: 1,964° C
Boiling Point: 3,695° C
Electron Affinity: 109.7 kJ/mol
Ionization, First: 719.7 kJ/mol
Radius Calculated: 173 pm
Hardness, Brinell: 1,100 MPa
Modulus, Bulk: 380 GPa
Density, STP: 12,450 kg/m3 W/mK
Conductivity, Thermal: 150 W/mK
Heat, Specific: 240 J/kgK
Abundance, Universe: 6.0E-8 %
Discovered: 18032
Series: Transition Metal
Atomic Number: 45
Energy Levels: 2, 8, 18, 16, 1
Atomic Weight: 102.90550 u
Electronegativity: 2.28
Melting Point: 1,964° C
Boiling Point: 3,695° C
Electron Affinity: 109.7 kJ/mol
Ionization, First: 719.7 kJ/mol
Radius Calculated: 173 pm
Hardness, Brinell: 1,100 MPa
Modulus, Bulk: 380 GPa
Density, STP: 12,450 kg/m3 W/mK
Conductivity, Thermal: 150 W/mK
Heat, Specific: 240 J/kgK
Abundance, Universe: 6.0E-8 %
Discovered: 1803
45
2
8
18
16
1
Rh
Rhodium
102.91
Element: Palladium
Series: Transition Metal
Atomic Number: 46
Energy Levels: 2, 8, 18, 18
Atomic Weight: 106.42 u
Electronegativity: 2.20
Melting Point: 1,554.9° C
Boiling Point: 2,963° C
Electron Affinity: 53.7 kJ/mol
Ionization, First: 804.4 kJ/mol
Radius Calculated: 169 pm
Hardness, Brinell: 37.3 MPa
Modulus, Bulk: 180 GPa
Density, STP: 12,023 kg/m3 W/mK
Conductivity, Thermal: 72 W/mK
Heat, Specific: 240 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 18032
Series: Transition Metal
Atomic Number: 46
Energy Levels: 2, 8, 18, 18
Atomic Weight: 106.42 u
Electronegativity: 2.20
Melting Point: 1,554.9° C
Boiling Point: 2,963° C
Electron Affinity: 53.7 kJ/mol
Ionization, First: 804.4 kJ/mol
Radius Calculated: 169 pm
Hardness, Brinell: 37.3 MPa
Modulus, Bulk: 180 GPa
Density, STP: 12,023 kg/m3 W/mK
Conductivity, Thermal: 72 W/mK
Heat, Specific: 240 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 1803
46
2
8
18
18
Pd
Palladium
106.42
Element: Silver
Series: Transition Metal
Atomic Number: 47
Energy Levels: 2, 8, 18, 18, 1
Atomic Weight: 107.8682 u
Electronegativity: 1.93
Melting Point: 961.780° C
Boiling Point: 2,162° C
Electron Affinity: 125.6 kJ/mol
Ionization, First: 731.0 kJ/mol
Radius Calculated: 165 pm
Hardness, Brinell: 24.5 MPa
Modulus, Bulk: 100 GPa
Density, STP: 10,490 kg/m3 W/mK
Conductivity, Thermal: 430 W/mK
Heat, Specific: 235 J/kgK
Abundance, Universe: 6.0E-8 %
Discovered: 3000 BCE2
Series: Transition Metal
Atomic Number: 47
Energy Levels: 2, 8, 18, 18, 1
Atomic Weight: 107.8682 u
Electronegativity: 1.93
Melting Point: 961.780° C
Boiling Point: 2,162° C
Electron Affinity: 125.6 kJ/mol
Ionization, First: 731.0 kJ/mol
Radius Calculated: 165 pm
Hardness, Brinell: 24.5 MPa
Modulus, Bulk: 100 GPa
Density, STP: 10,490 kg/m3 W/mK
Conductivity, Thermal: 430 W/mK
Heat, Specific: 235 J/kgK
Abundance, Universe: 6.0E-8 %
Discovered: 3000 BCE
47
2
8
18
18
1
Ag
Silver
107.87
Element: Cadmium
Series: Transition Metal
Atomic Number: 48
Energy Levels: 2, 8, 18, 18, 2
Atomic Weight: 112.414 u
Electronegativity: 1.69
Melting Point: 321.07° C
Boiling Point: 766.9° C
Electron Affinity: 0 kJ/mol
Ionization, First: 867.8 kJ/mol
Radius Calculated: 161 pm
Hardness, Brinell: 203 MPa
Modulus, Bulk: 42 GPa
Density, STP: 8,650 kg/m3 W/mK
Conductivity, Thermal: 97 W/mK
Heat, Specific: 230 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 18172
Series: Transition Metal
Atomic Number: 48
Energy Levels: 2, 8, 18, 18, 2
Atomic Weight: 112.414 u
Electronegativity: 1.69
Melting Point: 321.07° C
Boiling Point: 766.9° C
Electron Affinity: 0 kJ/mol
Ionization, First: 867.8 kJ/mol
Radius Calculated: 161 pm
Hardness, Brinell: 203 MPa
Modulus, Bulk: 42 GPa
Density, STP: 8,650 kg/m3 W/mK
Conductivity, Thermal: 97 W/mK
Heat, Specific: 230 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 1817
48
2
8
18
18
2
Cd
Cadmium
112.41
Element: Indium
Series: Post Transition Metal
Atomic Number: 49
Energy Levels: 2, 8, 18, 18, 3
Atomic Weight: 114.818 u
Electronegativity: 1.78
Melting Point: 156.60° C
Boiling Point: 2,072° C
Electron Affinity: 28.9 kJ/mol
Ionization, First: 558.3 kJ/mol
Radius Calculated: 156 pm
Hardness, Brinell: 8.83 MPa
Modulus, Bulk: N/A GPa
Density, STP: 7,310 kg/m3 W/mK
Conductivity, Thermal: 82 W/mK
Heat, Specific: 233 J/kgK
Abundance, Universe: 3.0E-8 %
Discovered: 18632
Series: Post Transition Metal
Atomic Number: 49
Energy Levels: 2, 8, 18, 18, 3
Atomic Weight: 114.818 u
Electronegativity: 1.78
Melting Point: 156.60° C
Boiling Point: 2,072° C
Electron Affinity: 28.9 kJ/mol
Ionization, First: 558.3 kJ/mol
Radius Calculated: 156 pm
Hardness, Brinell: 8.83 MPa
Modulus, Bulk: N/A GPa
Density, STP: 7,310 kg/m3 W/mK
Conductivity, Thermal: 82 W/mK
Heat, Specific: 233 J/kgK
Abundance, Universe: 3.0E-8 %
Discovered: 1863
49
2
8
18
18
3
In
Indium
114.82
Element: Tin
Series: Post Transition Metal
Atomic Number: 50
Energy Levels: 2, 8, 18, 18, 4
Atomic Weight: 118.710 u
Electronegativity: 1.96
Melting Point: 231.93° C
Boiling Point: 2,602° C
Electron Affinity: 107.3 kJ/mol
Ionization, First: 708.6 kJ/mol
Radius Calculated: 145 pm
Hardness, Brinell: 51 MPa
Modulus, Bulk: 58 GPa
Density, STP: 7,310 kg/m3 W/mK
Conductivity, Thermal: 67 W/mK
Heat, Specific: 217 J/kgK
Abundance, Universe: 4.0E-7 %
Discovered: 3000 BCE2
Series: Post Transition Metal
Atomic Number: 50
Energy Levels: 2, 8, 18, 18, 4
Atomic Weight: 118.710 u
Electronegativity: 1.96
Melting Point: 231.93° C
Boiling Point: 2,602° C
Electron Affinity: 107.3 kJ/mol
Ionization, First: 708.6 kJ/mol
Radius Calculated: 145 pm
Hardness, Brinell: 51 MPa
Modulus, Bulk: 58 GPa
Density, STP: 7,310 kg/m3 W/mK
Conductivity, Thermal: 67 W/mK
Heat, Specific: 217 J/kgK
Abundance, Universe: 4.0E-7 %
Discovered: 3000 BCE
50
2
8
18
18
4
Sn
Tin
118.71
Element: Iodine
Series: Reactive Nonmetal
Atomic Number: 53
Energy Levels: 2, 8, 18, 18, 7
Atomic Weight: 126.90447 u
Electronegativity: 2.66
Melting Point: 113.70° C
Boiling Point: 184.3° C
Electron Affinity: 295.2 kJ/mol
Ionization, First: 1,008.4 kJ/mol
Radius Calculated: 115 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 7.7 GPa
Density, STP: 4,940 kg/m3 W/mK
Conductivity, Thermal: 0.449 W/mK
Heat, Specific: 429.0 J/kgK
Abundance, Universe: 1.0E-7 %
Discovered: 18112
Series: Reactive Nonmetal
Atomic Number: 53
Energy Levels: 2, 8, 18, 18, 7
Atomic Weight: 126.90447 u
Electronegativity: 2.66
Melting Point: 113.70° C
Boiling Point: 184.3° C
Electron Affinity: 295.2 kJ/mol
Ionization, First: 1,008.4 kJ/mol
Radius Calculated: 115 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 7.7 GPa
Density, STP: 4,940 kg/m3 W/mK
Conductivity, Thermal: 0.449 W/mK
Heat, Specific: 429.0 J/kgK
Abundance, Universe: 1.0E-7 %
Discovered: 1811
53
2
8
18
18
7
I
Iodine
126.90
Element: Xenon
Series: Nobel Gas
Atomic Number: 54
Energy Levels: 2, 8, 18, 18, 8
Atomic Weight: 131.293 u
Electronegativity: 2.6
Melting Point: -111.8° C
Boiling Point: -108.0° C
Electron Affinity: 0 kJ/mol
Ionization, First: 1,170.4 kJ/mol
Radius Calculated: 108 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 5.9 kg/m3 W/mK
Conductivity, Thermal: 0.00565 W/mK
Heat, Specific: 158.32 J/kgK
Abundance, Universe: 1.0E-6 %
Discovered: 18982
Series: Nobel Gas
Atomic Number: 54
Energy Levels: 2, 8, 18, 18, 8
Atomic Weight: 131.293 u
Electronegativity: 2.6
Melting Point: -111.8° C
Boiling Point: -108.0° C
Electron Affinity: 0 kJ/mol
Ionization, First: 1,170.4 kJ/mol
Radius Calculated: 108 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 5.9 kg/m3 W/mK
Conductivity, Thermal: 0.00565 W/mK
Heat, Specific: 158.32 J/kgK
Abundance, Universe: 1.0E-6 %
Discovered: 1898
54
2
8
18
18
8
Xe
Xenon
131.29
6
Element: Caesium
Series: Alkali Metal
Atomic Number: 55
Energy Levels: 2, 8, 18, 18, 8, 1
Atomic Weight: 132.90545196 u
Electronegativity: 0.79
Melting Point: 28.440° C
Boiling Point: 671° C
Electron Affinity: 45.5 kJ/mol
Ionization, First: 375.7 kJ/mol
Radius Calculated: 298 pm
Hardness, Brinell: 0.14 MPa
Modulus, Bulk: 1.6 GPa
Density, STP: 1,879 kg/m3 W/mK
Conductivity, Thermal: 36 W/mK
Heat, Specific: 242 J/kgK
Abundance, Universe: 8.0E-8 %
Discovered: 18602
Series: Alkali Metal
Atomic Number: 55
Energy Levels: 2, 8, 18, 18, 8, 1
Atomic Weight: 132.90545196 u
Electronegativity: 0.79
Melting Point: 28.440° C
Boiling Point: 671° C
Electron Affinity: 45.5 kJ/mol
Ionization, First: 375.7 kJ/mol
Radius Calculated: 298 pm
Hardness, Brinell: 0.14 MPa
Modulus, Bulk: 1.6 GPa
Density, STP: 1,879 kg/m3 W/mK
Conductivity, Thermal: 36 W/mK
Heat, Specific: 242 J/kgK
Abundance, Universe: 8.0E-8 %
Discovered: 1860
55
2
8
18
18
8
1
Cs
Caesium
132.91
Element: Barium
Series: Alkaline Earth Metal
Atomic Number: 56
Energy Levels: 2, 8, 18, 18, 8, 2
Atomic Weight: 137.327 u
Electronegativity: 0.89
Melting Point: 730° C
Boiling Point: 1,870° C
Electron Affinity: 13.95 kJ/mol
Ionization, First: 502.9 kJ/mol
Radius Calculated: 253 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 9.6 GPa
Density, STP: 3,510 kg/m3 W/mK
Conductivity, Thermal: 18 W/mK
Heat, Specific: 205 J/kgK
Abundance, Universe: 1.00E-6 %
Discovered: 18082
Series: Alkaline Earth Metal
Atomic Number: 56
Energy Levels: 2, 8, 18, 18, 8, 2
Atomic Weight: 137.327 u
Electronegativity: 0.89
Melting Point: 730° C
Boiling Point: 1,870° C
Electron Affinity: 13.95 kJ/mol
Ionization, First: 502.9 kJ/mol
Radius Calculated: 253 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 9.6 GPa
Density, STP: 3,510 kg/m3 W/mK
Conductivity, Thermal: 18 W/mK
Heat, Specific: 205 J/kgK
Abundance, Universe: 1.00E-6 %
Discovered: 1808
56
2
8
18
18
8
2
Ba
Barium
137.33
Elements
57-71
Element: Hafnium
Series: Transition Metal
Atomic Number: 72
Energy Levels: 2, 8, 18, 32, 10, 2
Atomic Weight: 178.486 u
Electronegativity: 1.3
Melting Point: 2,233° C
Boiling Point: 4,603° C
Electron Affinity: 0 kJ/mol
Ionization, First: 658.5 kJ/mol
Radius Calculated: 208 pm
Hardness, Brinell: 1,700 MPa
Modulus, Bulk: 110 GPa
Density, STP: 13,310 kg/m3 W/mK
Conductivity, Thermal: 23 W/mK
Heat, Specific: 144 J/kgK
Abundance, Universe: 7.0E-8 %
Discovered: 19232
Series: Transition Metal
Atomic Number: 72
Energy Levels: 2, 8, 18, 32, 10, 2
Atomic Weight: 178.486 u
Electronegativity: 1.3
Melting Point: 2,233° C
Boiling Point: 4,603° C
Electron Affinity: 0 kJ/mol
Ionization, First: 658.5 kJ/mol
Radius Calculated: 208 pm
Hardness, Brinell: 1,700 MPa
Modulus, Bulk: 110 GPa
Density, STP: 13,310 kg/m3 W/mK
Conductivity, Thermal: 23 W/mK
Heat, Specific: 144 J/kgK
Abundance, Universe: 7.0E-8 %
Discovered: 1923
72
2
8
18
32
10
2
Hf
Hafnium
178.49
Element: Tantalum
Series: Transition Metal
Atomic Number: 73
Energy Levels: 2, 8, 18, 32, 11, 2
Atomic Weight: 180.94788 u
Electronegativity: 1.5
Melting Point: 3,017° C
Boiling Point: 5,458° C
Electron Affinity: 31 kJ/mol
Ionization, First: 761 kJ/mol
Radius Calculated: 200 pm
Hardness, Brinell: 800 MPa
Modulus, Bulk: 200 GPa
Density, STP: 16,650 kg/m3 W/mK
Conductivity, Thermal: 57 W/mK
Heat, Specific: 140 J/kgK
Abundance, Universe: 8.0E-9 %
Discovered: 18022
Series: Transition Metal
Atomic Number: 73
Energy Levels: 2, 8, 18, 32, 11, 2
Atomic Weight: 180.94788 u
Electronegativity: 1.5
Melting Point: 3,017° C
Boiling Point: 5,458° C
Electron Affinity: 31 kJ/mol
Ionization, First: 761 kJ/mol
Radius Calculated: 200 pm
Hardness, Brinell: 800 MPa
Modulus, Bulk: 200 GPa
Density, STP: 16,650 kg/m3 W/mK
Conductivity, Thermal: 57 W/mK
Heat, Specific: 140 J/kgK
Abundance, Universe: 8.0E-9 %
Discovered: 1802
73
2
8
18
32
11
2
Ta
Tantalum
180.95
Element: Tungsten
Series: Transition Metal
Atomic Number: 74
Energy Levels: 2, 8, 18, 32, 12, 2
Atomic Weight: 183.84 u
Electronegativity: 2.36
Melting Point: 3,422° C
Boiling Point: 5,555° C
Electron Affinity: 78.6 kJ/mol
Ionization, First: 770 kJ/mol
Radius Calculated: 193 pm
Hardness, Brinell: 2,570 MPa
Modulus, Bulk: 310 GPa
Density, STP: 19,250 kg/m3 W/mK
Conductivity, Thermal: 170 W/mK
Heat, Specific: 132 J/kgK
Abundance, Universe: 5.0E-8 %
Discovered: 17832
Series: Transition Metal
Atomic Number: 74
Energy Levels: 2, 8, 18, 32, 12, 2
Atomic Weight: 183.84 u
Electronegativity: 2.36
Melting Point: 3,422° C
Boiling Point: 5,555° C
Electron Affinity: 78.6 kJ/mol
Ionization, First: 770 kJ/mol
Radius Calculated: 193 pm
Hardness, Brinell: 2,570 MPa
Modulus, Bulk: 310 GPa
Density, STP: 19,250 kg/m3 W/mK
Conductivity, Thermal: 170 W/mK
Heat, Specific: 132 J/kgK
Abundance, Universe: 5.0E-8 %
Discovered: 1783
74
2
8
18
32
12
2
W
Tungsten
183.84
Element: Rhenium
Series: Transition Metal
Atomic Number: 75
Energy Levels: 2, 8, 18, 32, 13, 2
Atomic Weight: 186.207 u
Electronegativity: 1.9
Melting Point: 3,186° C
Boiling Point: 5,596° C
Electron Affinity: 14.5 kJ/mol
Ionization, First: 760 kJ/mol
Radius Calculated: 188 pm
Hardness, Brinell: 1,320 MPa
Modulus, Bulk: 370 GPa
Density, STP: 21,020 kg/m3 W/mK
Conductivity, Thermal: 48 W/mK
Heat, Specific: 137 J/kgK
Abundance, Universe: 2.0E-8 %
Discovered: 19252
Series: Transition Metal
Atomic Number: 75
Energy Levels: 2, 8, 18, 32, 13, 2
Atomic Weight: 186.207 u
Electronegativity: 1.9
Melting Point: 3,186° C
Boiling Point: 5,596° C
Electron Affinity: 14.5 kJ/mol
Ionization, First: 760 kJ/mol
Radius Calculated: 188 pm
Hardness, Brinell: 1,320 MPa
Modulus, Bulk: 370 GPa
Density, STP: 21,020 kg/m3 W/mK
Conductivity, Thermal: 48 W/mK
Heat, Specific: 137 J/kgK
Abundance, Universe: 2.0E-8 %
Discovered: 1925
75
2
8
18
32
13
2
Re
Rhenium
186.21
Element: Osmium
Series: Transition Metal
Atomic Number: 76
Energy Levels: 2, 8, 18, 32, 14, 2
Atomic Weight: 190.23 u
Electronegativity: 2.2
Melting Point: 3,033° C
Boiling Point: 5,012° C
Electron Affinity: 106.1 kJ/mol
Ionization, First: 840 kJ/mol
Radius Calculated: 185 pm
Hardness, Brinell: 3,920 MPa
Modulus, Bulk: N/A GPa
Density, STP: 22,590 kg/m3 W/mK
Conductivity, Thermal: 88 W/mK
Heat, Specific: 130 J/kgK
Abundance, Universe: 3.0E-7 %
Discovered: 18032
Series: Transition Metal
Atomic Number: 76
Energy Levels: 2, 8, 18, 32, 14, 2
Atomic Weight: 190.23 u
Electronegativity: 2.2
Melting Point: 3,033° C
Boiling Point: 5,012° C
Electron Affinity: 106.1 kJ/mol
Ionization, First: 840 kJ/mol
Radius Calculated: 185 pm
Hardness, Brinell: 3,920 MPa
Modulus, Bulk: N/A GPa
Density, STP: 22,590 kg/m3 W/mK
Conductivity, Thermal: 88 W/mK
Heat, Specific: 130 J/kgK
Abundance, Universe: 3.0E-7 %
Discovered: 1803
76
2
8
18
32
14
2
Os
Osmium
190.23
Element: Iridium
Series: Transition Metal
Atomic Number: 77
Energy Levels: 2, 8, 18, 32, 15, 2
Atomic Weight: 192.217 u
Electronegativity: 2.20
Melting Point: 2,466° C
Boiling Point: 4,428° C
Electron Affinity: 151 kJ/mol
Ionization, First: 880 kJ/mol
Radius Calculated: 180 pm
Hardness, Brinell: 1,670 MPa
Modulus, Bulk: 320 GPa
Density, STP: 22,560 kg/m3 W/mK
Conductivity, Thermal: 150 W/mK
Heat, Specific: 131 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 18032
Series: Transition Metal
Atomic Number: 77
Energy Levels: 2, 8, 18, 32, 15, 2
Atomic Weight: 192.217 u
Electronegativity: 2.20
Melting Point: 2,466° C
Boiling Point: 4,428° C
Electron Affinity: 151 kJ/mol
Ionization, First: 880 kJ/mol
Radius Calculated: 180 pm
Hardness, Brinell: 1,670 MPa
Modulus, Bulk: 320 GPa
Density, STP: 22,560 kg/m3 W/mK
Conductivity, Thermal: 150 W/mK
Heat, Specific: 131 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 1803
77
2
8
18
32
15
2
Ir
Iridium
192.22
Element: Platinum
Series: Transition Metal
Atomic Number: 78
Energy Levels: 2, 8, 18, 32, 17, 1
Atomic Weight: 195.084 u
Electronegativity: 2.28
Melting Point: 1,768.3° C
Boiling Point: 3,825° C
Electron Affinity: 205.3 kJ/mol
Ionization, First: 870 kJ/mol
Radius Calculated: 177 pm
Hardness, Brinell: 392 MPa
Modulus, Bulk: 230 GPa
Density, STP: 21,450 kg/m3 W/mK
Conductivity, Thermal: 72 W/mK
Heat, Specific: 133 J/kgK
Abundance, Universe: 5.0E-7 %
Discovered: 17352
Series: Transition Metal
Atomic Number: 78
Energy Levels: 2, 8, 18, 32, 17, 1
Atomic Weight: 195.084 u
Electronegativity: 2.28
Melting Point: 1,768.3° C
Boiling Point: 3,825° C
Electron Affinity: 205.3 kJ/mol
Ionization, First: 870 kJ/mol
Radius Calculated: 177 pm
Hardness, Brinell: 392 MPa
Modulus, Bulk: 230 GPa
Density, STP: 21,450 kg/m3 W/mK
Conductivity, Thermal: 72 W/mK
Heat, Specific: 133 J/kgK
Abundance, Universe: 5.0E-7 %
Discovered: 1735
78
2
8
18
32
17
1
Pt
Platinum
195.08
Element: Gold
Series: Transition Metal
Atomic Number: 79
Energy Levels: 2, 8, 18, 32, 18, 1
Atomic Weight: 196.966569 u
Electronegativity: 2.54
Melting Point: 1.064.18° C
Boiling Point: 2,856° C
Electron Affinity: 222.8 kJ/mol
Ionization, First: 890.1 kJ/mol
Radius Calculated: 174 pm
Hardness, Brinell: 25 MPa
Modulus, Bulk: 220 GPa
Density, STP: 19,300 kg/m3 W/mK
Conductivity, Thermal: 320 W/mK
Heat, Specific: 129.1 J/kgK
Abundance, Universe: 6.0E-8 %
Discovered: 2500 BCE2
Series: Transition Metal
Atomic Number: 79
Energy Levels: 2, 8, 18, 32, 18, 1
Atomic Weight: 196.966569 u
Electronegativity: 2.54
Melting Point: 1.064.18° C
Boiling Point: 2,856° C
Electron Affinity: 222.8 kJ/mol
Ionization, First: 890.1 kJ/mol
Radius Calculated: 174 pm
Hardness, Brinell: 25 MPa
Modulus, Bulk: 220 GPa
Density, STP: 19,300 kg/m3 W/mK
Conductivity, Thermal: 320 W/mK
Heat, Specific: 129.1 J/kgK
Abundance, Universe: 6.0E-8 %
Discovered: 2500 BCE
79
2
8
18
32
18
1
Au
Gold
196.97
Element: Mercury
Series: Transition Metal
Atomic Number: 80
Energy Levels: 2, 8, 18, 32, 18, 2
Atomic Weight: 200.59 u
Electronegativity: 2.0
Melting Point: -38.830° C
Boiling Point: 356.73° C
Electron Affinity: 0 kJ/mol
Ionization, First: 1,007.1 kJ/mol
Radius Calculated: 171 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 25 GPa
Density, STP: 13,534 kg/m3 W/mK
Conductivity, Thermal: 8.3 W/mK
Heat, Specific: 139.5 J/kgK
Abundance, Universe: 1.00E-7 %
Discovered: 1500 BCE2
Series: Transition Metal
Atomic Number: 80
Energy Levels: 2, 8, 18, 32, 18, 2
Atomic Weight: 200.59 u
Electronegativity: 2.0
Melting Point: -38.830° C
Boiling Point: 356.73° C
Electron Affinity: 0 kJ/mol
Ionization, First: 1,007.1 kJ/mol
Radius Calculated: 171 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 25 GPa
Density, STP: 13,534 kg/m3 W/mK
Conductivity, Thermal: 8.3 W/mK
Heat, Specific: 139.5 J/kgK
Abundance, Universe: 1.00E-7 %
Discovered: 1500 BCE
80
2
8
18
32
18
2
Hg
Mercury
200.59
Element: Thallium
Series: Post Transition Metal
Atomic Number: 81
Energy Levels: 2, 8, 18, 32, 18, 3
Atomic Weight: 204.38 u
Electronegativity: 1.62
Melting Point: 304° C
Boiling Point: 1,473° C
Electron Affinity: 19.2 kJ/mol
Ionization, First: 589.4 kJ/mol
Radius Calculated: 156 pm
Hardness, Brinell: 26.4 MPa
Modulus, Bulk: 43 GPa
Density, STP: 11,850 kg/m3 W/mK
Conductivity, Thermal: 46 W/mK
Heat, Specific: 129 J/kgK
Abundance, Universe: 5.0E-8 %
Discovered: 18612
Series: Post Transition Metal
Atomic Number: 81
Energy Levels: 2, 8, 18, 32, 18, 3
Atomic Weight: 204.38 u
Electronegativity: 1.62
Melting Point: 304° C
Boiling Point: 1,473° C
Electron Affinity: 19.2 kJ/mol
Ionization, First: 589.4 kJ/mol
Radius Calculated: 156 pm
Hardness, Brinell: 26.4 MPa
Modulus, Bulk: 43 GPa
Density, STP: 11,850 kg/m3 W/mK
Conductivity, Thermal: 46 W/mK
Heat, Specific: 129 J/kgK
Abundance, Universe: 5.0E-8 %
Discovered: 1861
81
2
8
18
32
18
3
Tl
Thallium
204.38
Element: Lead
Series: Post Transition Metal
Atomic Number: 82
Energy Levels: 2, 8, 18, 32, 18, 4
Atomic Weight: 207.2 u
Electronegativity: 2.33
Melting Point: 327.46° C
Boiling Point: 1,749° C
Electron Affinity: 35.1 kJ/mol
Ionization, First: 715.6 kJ/mol
Radius Calculated: 154 pm
Hardness, Brinell: 38.3 MPa
Modulus, Bulk: 46 GPa
Density, STP: 11,340 kg/m3 W/mK
Conductivity, Thermal: 35 W/mK
Heat, Specific: 127 J/kgK
Abundance, Universe: 1.00E-6 %
Discovered: 4000 BCE2
Series: Post Transition Metal
Atomic Number: 82
Energy Levels: 2, 8, 18, 32, 18, 4
Atomic Weight: 207.2 u
Electronegativity: 2.33
Melting Point: 327.46° C
Boiling Point: 1,749° C
Electron Affinity: 35.1 kJ/mol
Ionization, First: 715.6 kJ/mol
Radius Calculated: 154 pm
Hardness, Brinell: 38.3 MPa
Modulus, Bulk: 46 GPa
Density, STP: 11,340 kg/m3 W/mK
Conductivity, Thermal: 35 W/mK
Heat, Specific: 127 J/kgK
Abundance, Universe: 1.00E-6 %
Discovered: 4000 BCE
82
2
8
18
32
18
4
Pb
Lead
207.2
Element: Bismuth
Series: Post Transition Metal
Atomic Number: 83
Energy Levels: 2, 8, 18, 32, 18, 5
Atomic Weight: 208.98040 u
Electronegativity: 2.02
Melting Point: 271.3° C
Boiling Point: 1,564° C
Electron Affinity: 91.2 kJ/mol
Ionization, First: 703 kJ/mol
Radius Calculated: 143 pm
Hardness, Brinell: 94.2 MPa
Modulus, Bulk: 31 GPa
Density, STP: 9,780 kg/m3 W/mK
Conductivity, Thermal: 8 W/mK
Heat, Specific: 122 J/kgK
Abundance, Universe: 7.0E-8 %
Discovered: 14002
Series: Post Transition Metal
Atomic Number: 83
Energy Levels: 2, 8, 18, 32, 18, 5
Atomic Weight: 208.98040 u
Electronegativity: 2.02
Melting Point: 271.3° C
Boiling Point: 1,564° C
Electron Affinity: 91.2 kJ/mol
Ionization, First: 703 kJ/mol
Radius Calculated: 143 pm
Hardness, Brinell: 94.2 MPa
Modulus, Bulk: 31 GPa
Density, STP: 9,780 kg/m3 W/mK
Conductivity, Thermal: 8 W/mK
Heat, Specific: 122 J/kgK
Abundance, Universe: 7.0E-8 %
Discovered: 1400
83
2
8
18
32
18
5
Bi
Bismuth
208.98
Element: Polonium
Series: Post Transition Metal
Atomic Number: 84
Energy Levels: 2, 8, 18, 32, 18, 6
Atomic Weight: (209) u
Electronegativity: 2.0
Melting Point: 255° C
Boiling Point: 961.9° C
Electron Affinity: 183.3 kJ/mol
Ionization, First: 812.1 kJ/mol
Radius Calculated: 135 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 9,196 kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 18982
Series: Post Transition Metal
Atomic Number: 84
Energy Levels: 2, 8, 18, 32, 18, 6
Atomic Weight: (209) u
Electronegativity: 2.0
Melting Point: 255° C
Boiling Point: 961.9° C
Electron Affinity: 183.3 kJ/mol
Ionization, First: 812.1 kJ/mol
Radius Calculated: 135 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 9,196 kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1898
84
2
8
18
32
18
6
Po
Polonium
(209)
Element: Radon
Series: Nobel Gas
Atomic Number: 86
Energy Levels: 2, 8, 18, 32, 18, 8
Atomic Weight: (222) u
Electronegativity: N/A
Melting Point: -71.1° C
Boiling Point: -61.85° C
Electron Affinity: 0 kJ/mol
Ionization, First: 1,037 kJ/mol
Radius Calculated: 120 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 9.73 kg/m3 W/mK
Conductivity, Thermal: 0.00361 W/mK
Heat, Specific: 93.65 J/kgK
Abundance, Universe: 0 %
Discovered: 19002
Series: Nobel Gas
Atomic Number: 86
Energy Levels: 2, 8, 18, 32, 18, 8
Atomic Weight: (222) u
Electronegativity: N/A
Melting Point: -71.1° C
Boiling Point: -61.85° C
Electron Affinity: 0 kJ/mol
Ionization, First: 1,037 kJ/mol
Radius Calculated: 120 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 9.73 kg/m3 W/mK
Conductivity, Thermal: 0.00361 W/mK
Heat, Specific: 93.65 J/kgK
Abundance, Universe: 0 %
Discovered: 1900
86
2
8
18
32
18
8
Rn
Radon
(222)
7
Element: Francium
Series: Alkali Metal
Atomic Number: 87
Energy Levels: 2, 8, 18, 32, 18, 8, 1
Atomic Weight: (223) u
Electronegativity: 0.7
Melting Point: 20.9° C
Boiling Point: 650° C
Electron Affinity: N/A kJ/mol
Ionization, First: 380 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19392
Series: Alkali Metal
Atomic Number: 87
Energy Levels: 2, 8, 18, 32, 18, 8, 1
Atomic Weight: (223) u
Electronegativity: 0.7
Melting Point: 20.9° C
Boiling Point: 650° C
Electron Affinity: N/A kJ/mol
Ionization, First: 380 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1939
87
2
8
18
32
18
8
1
Fr
Francium
(223)
Element: Radium
Series: Alkaline Earth Metal
Atomic Number: 88
Energy Levels: 2, 8, 18, 32, 18, 8, 2
Atomic Weight: (226) u
Electronegativity: 0.9
Melting Point: 700° C
Boiling Point: 1,737° C
Electron Affinity: N/A kJ/mol
Ionization, First: 509.3 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 5,000 kg/m3 W/mK
Conductivity, Thermal: 19 W/mK
Heat, Specific: 92.0 J/kgK
Abundance, Universe: 0 %
Discovered: 18982
Series: Alkaline Earth Metal
Atomic Number: 88
Energy Levels: 2, 8, 18, 32, 18, 8, 2
Atomic Weight: (226) u
Electronegativity: 0.9
Melting Point: 700° C
Boiling Point: 1,737° C
Electron Affinity: N/A kJ/mol
Ionization, First: 509.3 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 5,000 kg/m3 W/mK
Conductivity, Thermal: 19 W/mK
Heat, Specific: 92.0 J/kgK
Abundance, Universe: 0 %
Discovered: 1898
88
2
8
18
32
18
8
2
Ra
Radium
(226)
Elements
89-103
Element: Rutherfordium
Series: Transition Metal
Atomic Number: 104
Energy Levels: 2, 8, 18, 32, 32, 10, 2
Atomic Weight: (267) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 580 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19642
Series: Transition Metal
Atomic Number: 104
Energy Levels: 2, 8, 18, 32, 32, 10, 2
Atomic Weight: (267) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 580 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1964
104
2
8
18
32
32
10
2
Rf
Rutherfordium
(267)
Element: Dubnium
Series: Transition Metal
Atomic Number: 105
Energy Levels: 2, 8, 18, 32, 32, 11, 2
Atomic Weight: (268) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19672
Series: Transition Metal
Atomic Number: 105
Energy Levels: 2, 8, 18, 32, 32, 11, 2
Atomic Weight: (268) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1967
105
2
8
18
32
32
11
2
Db
Dubnium
(268)
Element: Seaborgium
Series: Transition Metal
Atomic Number: 106
Energy Levels: 2, 8, 18, 32, 32, 12, 2
Atomic Weight: (269) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19742
Series: Transition Metal
Atomic Number: 106
Energy Levels: 2, 8, 18, 32, 32, 12, 2
Atomic Weight: (269) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1974
106
2
8
18
32
32
12
2
Sg
Seaborgium
(269)
Element: Bohrium
Series: Transition Metal
Atomic Number: 107
Energy Levels: 2, 8, 18, 32, 32, 13, 2
Atomic Weight: (270) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19812
Series: Transition Metal
Atomic Number: 107
Energy Levels: 2, 8, 18, 32, 32, 13, 2
Atomic Weight: (270) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1981
107
2
8
18
32
32
13
2
Bh
Bohrium
(270)
Element: Hassium
Series: Transition Metal
Atomic Number: 108
Energy Levels: 2, 8, 18, 32, 32, 14, 2
Atomic Weight: (277) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19842
Series: Transition Metal
Atomic Number: 108
Energy Levels: 2, 8, 18, 32, 32, 14, 2
Atomic Weight: (277) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1984
108
2
8
18
32
32
14
2
Hs
Hassium
(277)
Element: Meitnerium
Series: N/A
Atomic Number: 109
Energy Levels: 2, 8, 18, 32, 32, 15, 2
Atomic Weight: (278) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19822
Series: N/A
Atomic Number: 109
Energy Levels: 2, 8, 18, 32, 32, 15, 2
Atomic Weight: (278) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1982
109
2
8
18
32
32
15
2
Mt
Meitnerium
(278)
Element: Darmstadtium
Series: N/A
Atomic Number: 110
Energy Levels: 2, 8, 18, 32, 32, 17, 1
Atomic Weight: (281) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19942
Series: N/A
Atomic Number: 110
Energy Levels: 2, 8, 18, 32, 32, 17, 1
Atomic Weight: (281) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1994
110
2
8
18
32
32
17
1
Ds
Darmstadtium
(281)
Element: Roentgenium
Series: N/A
Atomic Number: 111
Energy Levels: 2, 8, 18, 32, 32, 17, 2
Atomic Weight: (282) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19942
Series: N/A
Atomic Number: 111
Energy Levels: 2, 8, 18, 32, 32, 17, 2
Atomic Weight: (282) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1994
111
2
8
18
32
32
17
2
Rg
Roentgenium
(282)
Element: Copernicium
Series: N/A
Atomic Number: 112
Energy Levels: 2, 8, 18, 32, 32, 18, 2
Atomic Weight: (285) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19962
Series: N/A
Atomic Number: 112
Energy Levels: 2, 8, 18, 32, 32, 18, 2
Atomic Weight: (285) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1996
112
2
8
18
32
32
18
2
Cn
Copernicium
(285)
Element: Nihonium
Series: N/A
Atomic Number: 113
Energy Levels: 2, 8, 18, 32, 32, 18, 3
Atomic Weight: (286) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 20042
Series: N/A
Atomic Number: 113
Energy Levels: 2, 8, 18, 32, 32, 18, 3
Atomic Weight: (286) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 2004
113
2
8
18
32
32
18
3
Nh
Nihonium
(286)
Element: Flerovium
Series: N/A
Atomic Number: 114
Energy Levels: 2, 8, 18, 32, 32, 18, 4
Atomic Weight: (289) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19982
Series: N/A
Atomic Number: 114
Energy Levels: 2, 8, 18, 32, 32, 18, 4
Atomic Weight: (289) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1998
114
2
8
18
32
32
18
4
Fl
Flerovium
(289)
Element: Moscovium
Series: N/A
Atomic Number: 115
Energy Levels: 2, 8, 18, 32, 32, 18, 5
Atomic Weight: (290) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 20042
Series: N/A
Atomic Number: 115
Energy Levels: 2, 8, 18, 32, 32, 18, 5
Atomic Weight: (290) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 2004
115
2
8
18
32
32
18
5
Mc
Moscovium
(290)
Element: Livermorium
Series: N/A
Atomic Number: 116
Energy Levels: 2, 8, 18, 32, 32, 18, 6
Atomic Weight: (293) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 20002
Series: N/A
Atomic Number: 116
Energy Levels: 2, 8, 18, 32, 32, 18, 6
Atomic Weight: (293) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 2000
116
2
8
18
32
32
18
6
Lv
Livermorium
(293)
Element: Tennessine
Series: N/A
Atomic Number: 117
Energy Levels: 2, 8, 18, 32, 32, 18, 7
Atomic Weight: (294) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 20102
Series: N/A
Atomic Number: 117
Energy Levels: 2, 8, 18, 32, 32, 18, 7
Atomic Weight: (294) u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 2010
117
2
8
18
32
32
18
7
Ts
Tennessine
(294)
Element: Oganesson
Series: N/A
Atomic Number: 118
Energy Levels: 2, 8, 18, 32, 32, 18, 8
Atomic Weight: 294 u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 20062
Series: N/A
Atomic Number: 118
Energy Levels: 2, 8, 18, 32, 32, 18, 8
Atomic Weight: 294 u
Electronegativity: N/A
Melting Point: N/A° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: N/A kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 2006
118
2
8
18
32
32
18
8
Og
Oganesson
294
↓
6
Element: Lanthanum
Series: Lanthanoids
Atomic Number: 57
Energy Levels: 2, 8, 18, 18, 9, 2
Atomic Weight: 138.90547 u
Electronegativity: 1.10
Melting Point: 919.9° C
Boiling Point: 3,464° C
Electron Affinity: 48 kJ/mol
Ionization, First: 538.1 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: 363 MPa
Modulus, Bulk: 28 GPa
Density, STP: 6,146 kg/m3 W/mK
Conductivity, Thermal: 13 W/mK
Heat, Specific: 195 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 18392
Series: Lanthanoids
Atomic Number: 57
Energy Levels: 2, 8, 18, 18, 9, 2
Atomic Weight: 138.90547 u
Electronegativity: 1.10
Melting Point: 919.9° C
Boiling Point: 3,464° C
Electron Affinity: 48 kJ/mol
Ionization, First: 538.1 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: 363 MPa
Modulus, Bulk: 28 GPa
Density, STP: 6,146 kg/m3 W/mK
Conductivity, Thermal: 13 W/mK
Heat, Specific: 195 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 1839
57
2
8
18
18
9
2
La
Lanthanum
138.91
Element: Cerium
Series: Lanthanoids
Atomic Number: 58
Energy Levels: 2, 8, 18, 19, 9, 2
Atomic Weight: 140.116 u
Electronegativity: 1.12
Melting Point: 797.9° C
Boiling Point: 3,360° C
Electron Affinity: 50 kJ/mol
Ionization, First: 534.4 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: 412 MPa
Modulus, Bulk: 22 GPa
Density, STP: 6,689 kg/m3 W/mK
Conductivity, Thermal: 11 W/mK
Heat, Specific: 192 J/kgK
Abundance, Universe: 1.00E-6 %
Discovered: 18032
Series: Lanthanoids
Atomic Number: 58
Energy Levels: 2, 8, 18, 19, 9, 2
Atomic Weight: 140.116 u
Electronegativity: 1.12
Melting Point: 797.9° C
Boiling Point: 3,360° C
Electron Affinity: 50 kJ/mol
Ionization, First: 534.4 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: 412 MPa
Modulus, Bulk: 22 GPa
Density, STP: 6,689 kg/m3 W/mK
Conductivity, Thermal: 11 W/mK
Heat, Specific: 192 J/kgK
Abundance, Universe: 1.00E-6 %
Discovered: 1803
58
2
8
18
19
9
2
Ce
Cerium
140.12
Element: Praseodymium
Series: Lanthanoids
Atomic Number: 59
Energy Levels: 2, 8, 18, 21, 8, 2
Atomic Weight: 140.90766 u
Electronegativity: 1.13
Melting Point: 930.9° C
Boiling Point: 3,290° C
Electron Affinity: 50 kJ/mol
Ionization, First: 527 kJ/mol
Radius Calculated: 247 pm
Hardness, Brinell: 481 MPa
Modulus, Bulk: 29 GPa
Density, STP: 6,640 kg/m3 W/mK
Conductivity, Thermal: 13 W/mK
Heat, Specific: 193 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 18852
Series: Lanthanoids
Atomic Number: 59
Energy Levels: 2, 8, 18, 21, 8, 2
Atomic Weight: 140.90766 u
Electronegativity: 1.13
Melting Point: 930.9° C
Boiling Point: 3,290° C
Electron Affinity: 50 kJ/mol
Ionization, First: 527 kJ/mol
Radius Calculated: 247 pm
Hardness, Brinell: 481 MPa
Modulus, Bulk: 29 GPa
Density, STP: 6,640 kg/m3 W/mK
Conductivity, Thermal: 13 W/mK
Heat, Specific: 193 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 1885
59
2
8
18
21
8
2
Pr
Praseodymium
140.91
Element: Neodymium
Series: Lanthanoids
Atomic Number: 60
Energy Levels: 2, 8, 18, 22, 8, 2
Atomic Weight: 144.242 u
Electronegativity: 1.14
Melting Point: 1,021° C
Boiling Point: 3,100° C
Electron Affinity: 50 kJ/mol
Ionization, First: 533.1 kJ/mol
Radius Calculated: 206 pm
Hardness, Brinell: 265 MPa
Modulus, Bulk: 32 GPa
Density, STP: 7,010 kg/m3 W/mK
Conductivity, Thermal: 17 W/mK
Heat, Specific: 190 J/kgK
Abundance, Universe: 1.00E-6 %
Discovered: 18852
Series: Lanthanoids
Atomic Number: 60
Energy Levels: 2, 8, 18, 22, 8, 2
Atomic Weight: 144.242 u
Electronegativity: 1.14
Melting Point: 1,021° C
Boiling Point: 3,100° C
Electron Affinity: 50 kJ/mol
Ionization, First: 533.1 kJ/mol
Radius Calculated: 206 pm
Hardness, Brinell: 265 MPa
Modulus, Bulk: 32 GPa
Density, STP: 7,010 kg/m3 W/mK
Conductivity, Thermal: 17 W/mK
Heat, Specific: 190 J/kgK
Abundance, Universe: 1.00E-6 %
Discovered: 1885
60
2
8
18
22
8
2
Nd
Neodymium
144.24
Element: Promethium
Series: Lanthanoids
Atomic Number: 61
Energy Levels: 2, 8, 18, 23, 8, 2
Atomic Weight: (145) u
Electronegativity: N/A
Melting Point: 1,100° C
Boiling Point: 3,000° C
Electron Affinity: 50 kJ/mol
Ionization, First: 540 kJ/mol
Radius Calculated: 205 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 33 GPa
Density, STP: 7,264 kg/m3 W/mK
Conductivity, Thermal: 15 W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19452
Series: Lanthanoids
Atomic Number: 61
Energy Levels: 2, 8, 18, 23, 8, 2
Atomic Weight: (145) u
Electronegativity: N/A
Melting Point: 1,100° C
Boiling Point: 3,000° C
Electron Affinity: 50 kJ/mol
Ionization, First: 540 kJ/mol
Radius Calculated: 205 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 33 GPa
Density, STP: 7,264 kg/m3 W/mK
Conductivity, Thermal: 15 W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1945
61
2
8
18
23
8
2
Pm
Promethium
(145)
Element: Samarium
Series: Lanthanoids
Atomic Number: 62
Energy Levels: 2, 8, 18, 24, 8, 2
Atomic Weight: 150.36 u
Electronegativity: 1.17
Melting Point: 1.072° C
Boiling Point: 1,803° C
Electron Affinity: 50 kJ/mol
Ionization, First: 544.5 kJ/mol
Radius Calculated: 238 pm
Hardness, Brinell: 441 MPa
Modulus, Bulk: 38 GPa
Density, STP: 7,353 kg/m3 W/mK
Conductivity, Thermal: 13 W/mK
Heat, Specific: 196 J/kgK
Abundance, Universe: 5.0E-7 %
Discovered: 18792
Series: Lanthanoids
Atomic Number: 62
Energy Levels: 2, 8, 18, 24, 8, 2
Atomic Weight: 150.36 u
Electronegativity: 1.17
Melting Point: 1.072° C
Boiling Point: 1,803° C
Electron Affinity: 50 kJ/mol
Ionization, First: 544.5 kJ/mol
Radius Calculated: 238 pm
Hardness, Brinell: 441 MPa
Modulus, Bulk: 38 GPa
Density, STP: 7,353 kg/m3 W/mK
Conductivity, Thermal: 13 W/mK
Heat, Specific: 196 J/kgK
Abundance, Universe: 5.0E-7 %
Discovered: 1879
62
2
8
18
24
8
2
Sm
Samarium
150.36
Element: Europium
Series: Lanthanoids
Atomic Number: 63
Energy Levels: 2, 8, 18, 25, 8, 2
Atomic Weight: 151.964 u
Electronegativity: N/A
Melting Point: 821.9° C
Boiling Point: 1,500° C
Electron Affinity: 50 kJ/mol
Ionization, First: 547.1 kJ/mol
Radius Calculated: 231 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 8.3 GPa
Density, STP: 5,244 kg/m3 W/mK
Conductivity, Thermal: 14 W/mK
Heat, Specific: 182 J/kgK
Abundance, Universe: 5.0E-8 %
Discovered: 19012
Series: Lanthanoids
Atomic Number: 63
Energy Levels: 2, 8, 18, 25, 8, 2
Atomic Weight: 151.964 u
Electronegativity: N/A
Melting Point: 821.9° C
Boiling Point: 1,500° C
Electron Affinity: 50 kJ/mol
Ionization, First: 547.1 kJ/mol
Radius Calculated: 231 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 8.3 GPa
Density, STP: 5,244 kg/m3 W/mK
Conductivity, Thermal: 14 W/mK
Heat, Specific: 182 J/kgK
Abundance, Universe: 5.0E-8 %
Discovered: 1901
63
2
8
18
25
8
2
Eu
Europium
151.96
Element: Gadolinium
Series: Lanthanoids
Atomic Number: 64
Energy Levels: 2, 8, 18, 25, 9, 2
Atomic Weight: 157.25 u
Electronegativity: 1.20
Melting Point: 1,313° C
Boiling Point: 3,250° C
Electron Affinity: 50 kJ/mol
Ionization, First: 593.4 kJ/mol
Radius Calculated: 233 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 38 GPa
Density, STP: 7,901 kg/m3 W/mK
Conductivity, Thermal: 11 W/mK
Heat, Specific: 240 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 18802
Series: Lanthanoids
Atomic Number: 64
Energy Levels: 2, 8, 18, 25, 9, 2
Atomic Weight: 157.25 u
Electronegativity: 1.20
Melting Point: 1,313° C
Boiling Point: 3,250° C
Electron Affinity: 50 kJ/mol
Ionization, First: 593.4 kJ/mol
Radius Calculated: 233 pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: 38 GPa
Density, STP: 7,901 kg/m3 W/mK
Conductivity, Thermal: 11 W/mK
Heat, Specific: 240 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 1880
64
2
8
18
25
9
2
Gd
Gadolinium
157.25
Element: Terbium
Series: Lanthanoids
Atomic Number: 65
Energy Levels: 2, 8, 18, 27, 8, 2
Atomic Weight: 158.92535 u
Electronegativity: N/A
Melting Point: 1,356° C
Boiling Point: 3,230° C
Electron Affinity: 50 kJ/mol
Ionization, First: 565.8 kJ/mol
Radius Calculated: 225 pm
Hardness, Brinell: 677 MPa
Modulus, Bulk: 38.7 GPa
Density, STP: 8,219 kg/m3 W/mK
Conductivity, Thermal: 11 W/mK
Heat, Specific: 182 J/kgK
Abundance, Universe: 5.0E-8 %
Discovered: 18432
Series: Lanthanoids
Atomic Number: 65
Energy Levels: 2, 8, 18, 27, 8, 2
Atomic Weight: 158.92535 u
Electronegativity: N/A
Melting Point: 1,356° C
Boiling Point: 3,230° C
Electron Affinity: 50 kJ/mol
Ionization, First: 565.8 kJ/mol
Radius Calculated: 225 pm
Hardness, Brinell: 677 MPa
Modulus, Bulk: 38.7 GPa
Density, STP: 8,219 kg/m3 W/mK
Conductivity, Thermal: 11 W/mK
Heat, Specific: 182 J/kgK
Abundance, Universe: 5.0E-8 %
Discovered: 1843
65
2
8
18
27
8
2
Tb
Terbium
158.93
Element: Dysprosium
Series: Lanthanoids
Atomic Number: 66
Energy Levels: 2, 8, 18, 28, 8, 2
Atomic Weight: 162.500 u
Electronegativity: 1.22
Melting Point: 1,412° C
Boiling Point: 2,567° C
Electron Affinity: 50 kJ/mol
Ionization, First: 573.0 kJ/mol
Radius Calculated: 228 pm
Hardness, Brinell: 500 MPa
Modulus, Bulk: 41 GPa
Density, STP: 8,551 kg/m3 W/mK
Conductivity, Thermal: 11 W/mK
Heat, Specific: 167 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 18862
Series: Lanthanoids
Atomic Number: 66
Energy Levels: 2, 8, 18, 28, 8, 2
Atomic Weight: 162.500 u
Electronegativity: 1.22
Melting Point: 1,412° C
Boiling Point: 2,567° C
Electron Affinity: 50 kJ/mol
Ionization, First: 573.0 kJ/mol
Radius Calculated: 228 pm
Hardness, Brinell: 500 MPa
Modulus, Bulk: 41 GPa
Density, STP: 8,551 kg/m3 W/mK
Conductivity, Thermal: 11 W/mK
Heat, Specific: 167 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 1886
66
2
8
18
28
8
2
Dy
Dysprosium
162.50
Element: Holmium
Series: Lanthanoids
Atomic Number: 67
Energy Levels: 2, 8, 18, 29, 8, 2
Atomic Weight: 164.93033 u
Electronegativity: 1.23
Melting Point: 1,474° C
Boiling Point: 2,700° C
Electron Affinity: 50 kJ/mol
Ionization, First: 581.0 kJ/mol
Radius Calculated: 226 pm
Hardness, Brinell: 746 MPa
Modulus, Bulk: 40 GPa
Density, STP: 8,795 kg/m3 W/mK
Conductivity, Thermal: 16 W/mK
Heat, Specific: 165 J/kgK
Abundance, Universe: 5.0E-8 %
Discovered: 18782
Series: Lanthanoids
Atomic Number: 67
Energy Levels: 2, 8, 18, 29, 8, 2
Atomic Weight: 164.93033 u
Electronegativity: 1.23
Melting Point: 1,474° C
Boiling Point: 2,700° C
Electron Affinity: 50 kJ/mol
Ionization, First: 581.0 kJ/mol
Radius Calculated: 226 pm
Hardness, Brinell: 746 MPa
Modulus, Bulk: 40 GPa
Density, STP: 8,795 kg/m3 W/mK
Conductivity, Thermal: 16 W/mK
Heat, Specific: 165 J/kgK
Abundance, Universe: 5.0E-8 %
Discovered: 1878
67
2
8
18
29
8
2
Ho
Holmium
164.93
Element: Erbium
Series: Lanthanoids
Atomic Number: 68
Energy Levels: 2, 8, 18, 30, 8, 2
Atomic Weight: 167.259 u
Electronegativity: 1.24
Melting Point: 1,497° C
Boiling Point: 2,868° C
Electron Affinity: 50 kJ/mol
Ionization, First: 589.3 kJ/mol
Radius Calculated: 226 pm
Hardness, Brinell: 814 MPa
Modulus, Bulk: 44 GPa
Density, STP: 9,066 kg/m3 W/mK
Conductivity, Thermal: 15 W/mK
Heat, Specific: 168 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 18422
Series: Lanthanoids
Atomic Number: 68
Energy Levels: 2, 8, 18, 30, 8, 2
Atomic Weight: 167.259 u
Electronegativity: 1.24
Melting Point: 1,497° C
Boiling Point: 2,868° C
Electron Affinity: 50 kJ/mol
Ionization, First: 589.3 kJ/mol
Radius Calculated: 226 pm
Hardness, Brinell: 814 MPa
Modulus, Bulk: 44 GPa
Density, STP: 9,066 kg/m3 W/mK
Conductivity, Thermal: 15 W/mK
Heat, Specific: 168 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 1842
68
2
8
18
30
8
2
Er
Erbium
167.26
Element: Thulium
Series: Lanthanoids
Atomic Number: 69
Energy Levels: 2, 8, 18, 31, 8, 2
Atomic Weight: 168.93422 u
Electronegativity: 1.25
Melting Point: 1,545° C
Boiling Point: 1,950° C
Electron Affinity: 50 kJ/mol
Ionization, First: 596.7 kJ/mol
Radius Calculated: 222 pm
Hardness, Brinell: 471 MPa
Modulus, Bulk: 45 GPa
Density, STP: 9,320 kg/m3 W/mK
Conductivity, Thermal: 17 W/mK
Heat, Specific: 160 J/kgK
Abundance, Universe: 1.00E-8 %
Discovered: 18792
Series: Lanthanoids
Atomic Number: 69
Energy Levels: 2, 8, 18, 31, 8, 2
Atomic Weight: 168.93422 u
Electronegativity: 1.25
Melting Point: 1,545° C
Boiling Point: 1,950° C
Electron Affinity: 50 kJ/mol
Ionization, First: 596.7 kJ/mol
Radius Calculated: 222 pm
Hardness, Brinell: 471 MPa
Modulus, Bulk: 45 GPa
Density, STP: 9,320 kg/m3 W/mK
Conductivity, Thermal: 17 W/mK
Heat, Specific: 160 J/kgK
Abundance, Universe: 1.00E-8 %
Discovered: 1879
69
2
8
18
31
8
2
Tm
Thulium
168.93
Element: Ytterbium
Series: Lanthanoids
Atomic Number: 70
Energy Levels: 2, 8, 18, 32, 8, 2
Atomic Weight: 173.045 u
Electronegativity: N/A
Melting Point: 818.9° C
Boiling Point: 1,196° C
Electron Affinity: 50 kJ/mol
Ionization, First: 603.4 kJ/mol
Radius Calculated: 222 pm
Hardness, Brinell: 343 MPa
Modulus, Bulk: 31 GPa
Density, STP: 6,570 kg/m3 W/mK
Conductivity, Thermal: 39 W/mK
Heat, Specific: 154 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 18782
Series: Lanthanoids
Atomic Number: 70
Energy Levels: 2, 8, 18, 32, 8, 2
Atomic Weight: 173.045 u
Electronegativity: N/A
Melting Point: 818.9° C
Boiling Point: 1,196° C
Electron Affinity: 50 kJ/mol
Ionization, First: 603.4 kJ/mol
Radius Calculated: 222 pm
Hardness, Brinell: 343 MPa
Modulus, Bulk: 31 GPa
Density, STP: 6,570 kg/m3 W/mK
Conductivity, Thermal: 39 W/mK
Heat, Specific: 154 J/kgK
Abundance, Universe: 2.0E-7 %
Discovered: 1878
70
2
8
18
32
8
2
Yb
Ytterbium
173.05
Element: Lutetium
Series: Lanthanoids
Atomic Number: 71
Energy Levels: 2, 8, 18, 32, 9, 2
Atomic Weight: 174.9668 u
Electronegativity: 1.27
Melting Point: 1,663° C
Boiling Point: 3,402° C
Electron Affinity: 50 kJ/mol
Ionization, First: 523.5 kJ/mol
Radius Calculated: 217 pm
Hardness, Brinell: 893 MPa
Modulus, Bulk: 48 GPa
Density, STP: 9,841 kg/m3 W/mK
Conductivity, Thermal: 16 W/mK
Heat, Specific: 154 J/kgK
Abundance, Universe: 1.00E-8 %
Discovered: 19072
Series: Lanthanoids
Atomic Number: 71
Energy Levels: 2, 8, 18, 32, 9, 2
Atomic Weight: 174.9668 u
Electronegativity: 1.27
Melting Point: 1,663° C
Boiling Point: 3,402° C
Electron Affinity: 50 kJ/mol
Ionization, First: 523.5 kJ/mol
Radius Calculated: 217 pm
Hardness, Brinell: 893 MPa
Modulus, Bulk: 48 GPa
Density, STP: 9,841 kg/m3 W/mK
Conductivity, Thermal: 16 W/mK
Heat, Specific: 154 J/kgK
Abundance, Universe: 1.00E-8 %
Discovered: 1907
71
2
8
18
32
9
2
Lu
Lutetium
174.97
7
Element: Actinium
Series: Actinoids
Atomic Number: 89
Energy Levels: 2, 8, 18, 32, 18, 9, 2
Atomic Weight: (227) u
Electronegativity: 1.1
Melting Point: 1,050° C
Boiling Point: 3,200° C
Electron Affinity: N/A kJ/mol
Ionization, First: 499 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 10,070 kg/m3 W/mK
Conductivity, Thermal: 12 W/mK
Heat, Specific: 120 J/kgK
Abundance, Universe: 0 %
Discovered: 18992
Series: Actinoids
Atomic Number: 89
Energy Levels: 2, 8, 18, 32, 18, 9, 2
Atomic Weight: (227) u
Electronegativity: 1.1
Melting Point: 1,050° C
Boiling Point: 3,200° C
Electron Affinity: N/A kJ/mol
Ionization, First: 499 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 10,070 kg/m3 W/mK
Conductivity, Thermal: 12 W/mK
Heat, Specific: 120 J/kgK
Abundance, Universe: 0 %
Discovered: 1899
89
2
8
18
32
9
2
Ac
Actinium
(227)
Element: Thorium
Series: Actinoids
Atomic Number: 90
Energy Levels: 2, 8, 18, 32, 18, 10, 2
Atomic Weight: 232.0377 u
Electronegativity: 1.3
Melting Point: 1,750° C
Boiling Point: 4,820° C
Electron Affinity: N/A kJ/mol
Ionization, First: 587 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: 400 MPa
Modulus, Bulk: 54 GPa
Density, STP: 11,724 kg/m3 W/mK
Conductivity, Thermal: 54 W/mK
Heat, Specific: 118 J/kgK
Abundance, Universe: 4.0E-8 %
Discovered: 18292
Series: Actinoids
Atomic Number: 90
Energy Levels: 2, 8, 18, 32, 18, 10, 2
Atomic Weight: 232.0377 u
Electronegativity: 1.3
Melting Point: 1,750° C
Boiling Point: 4,820° C
Electron Affinity: N/A kJ/mol
Ionization, First: 587 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: 400 MPa
Modulus, Bulk: 54 GPa
Density, STP: 11,724 kg/m3 W/mK
Conductivity, Thermal: 54 W/mK
Heat, Specific: 118 J/kgK
Abundance, Universe: 4.0E-8 %
Discovered: 1829
90
2
8
18
32
10
2
Th
Thorium
232.04
Element: Protactinium
Series: Actinoids
Atomic Number: 91
Energy Levels: 2, 8, 18, 32, 20, 9, 2
Atomic Weight: 231.03588 u
Electronegativity: 1.5
Melting Point: 1,572° C
Boiling Point: 4,000° C
Electron Affinity: N/A kJ/mol
Ionization, First: 568 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 15,370 kg/m3 W/mK
Conductivity, Thermal: 47 W/mK
Heat, Specific: 99.1 J/kgK
Abundance, Universe: 0 %
Discovered: 19132
Series: Actinoids
Atomic Number: 91
Energy Levels: 2, 8, 18, 32, 20, 9, 2
Atomic Weight: 231.03588 u
Electronegativity: 1.5
Melting Point: 1,572° C
Boiling Point: 4,000° C
Electron Affinity: N/A kJ/mol
Ionization, First: 568 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 15,370 kg/m3 W/mK
Conductivity, Thermal: 47 W/mK
Heat, Specific: 99.1 J/kgK
Abundance, Universe: 0 %
Discovered: 1913
91
2
8
18
32
20
9
2
Pa
Protactinium
231.04
Element: Uranium
Series: Actinoids
Atomic Number: 92
Energy Levels: 2, 8, 18, 32, 21, 9, 2
Atomic Weight: 238.02891 u
Electronegativity: 1.38
Melting Point: 1,135° C
Boiling Point: 3,900° C
Electron Affinity: N/A kJ/mol
Ionization, First: 597.6 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: 2,400 MPa
Modulus, Bulk: 100 GPa
Density, STP: 19,050 kg/m3 W/mK
Conductivity, Thermal: 27 W/mK
Heat, Specific: 116 J/kgK
Abundance, Universe: 2.0E-8 %
Discovered: 17892
Series: Actinoids
Atomic Number: 92
Energy Levels: 2, 8, 18, 32, 21, 9, 2
Atomic Weight: 238.02891 u
Electronegativity: 1.38
Melting Point: 1,135° C
Boiling Point: 3,900° C
Electron Affinity: N/A kJ/mol
Ionization, First: 597.6 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: 2,400 MPa
Modulus, Bulk: 100 GPa
Density, STP: 19,050 kg/m3 W/mK
Conductivity, Thermal: 27 W/mK
Heat, Specific: 116 J/kgK
Abundance, Universe: 2.0E-8 %
Discovered: 1789
92
2
8
18
32
21
9
2
U
Uranium
238.03
Element: Neptunium
Series: Actinoids
Atomic Number: 93
Energy Levels: 2, 8, 18, 32, 22, 9, 2
Atomic Weight: (237) u
Electronegativity: 1.36
Melting Point: 644° C
Boiling Point: 4,000° C
Electron Affinity: N/A kJ/mol
Ionization, First: 604.5 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 20,450 kg/m3 W/mK
Conductivity, Thermal: 6 W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19402
Series: Actinoids
Atomic Number: 93
Energy Levels: 2, 8, 18, 32, 22, 9, 2
Atomic Weight: (237) u
Electronegativity: 1.36
Melting Point: 644° C
Boiling Point: 4,000° C
Electron Affinity: N/A kJ/mol
Ionization, First: 604.5 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 20,450 kg/m3 W/mK
Conductivity, Thermal: 6 W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1940
93
2
8
18
32
22
9
2
Np
Neptunium
(237)
Element: Plutonium
Series: Actinoids
Atomic Number: 94
Energy Levels: 2, 8, 18, 32, 24, 8, 2
Atomic Weight: (244) u
Electronegativity: 1.28
Melting Point: 640° C
Boiling Point: 3,230° C
Electron Affinity: N/A kJ/mol
Ionization, First: 584.7 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 19,816 kg/m3 W/mK
Conductivity, Thermal: 6 W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19402
Series: Actinoids
Atomic Number: 94
Energy Levels: 2, 8, 18, 32, 24, 8, 2
Atomic Weight: (244) u
Electronegativity: 1.28
Melting Point: 640° C
Boiling Point: 3,230° C
Electron Affinity: N/A kJ/mol
Ionization, First: 584.7 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 19,816 kg/m3 W/mK
Conductivity, Thermal: 6 W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1940
94
2
8
18
32
24
8
2
Pu
Plutonium
(244)
Element: Americium
Series: Actinoids
Atomic Number: 95
Energy Levels: 2, 8, 18, 32, 25, 8, 2
Atomic Weight: (243) u
Electronegativity: 1.3
Melting Point: 1,176° C
Boiling Point: 2,011° C
Electron Affinity: N/A kJ/mol
Ionization, First: 578 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 13,670 kg/m3 W/mK
Conductivity, Thermal: 10 W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19442
Series: Actinoids
Atomic Number: 95
Energy Levels: 2, 8, 18, 32, 25, 8, 2
Atomic Weight: (243) u
Electronegativity: 1.3
Melting Point: 1,176° C
Boiling Point: 2,011° C
Electron Affinity: N/A kJ/mol
Ionization, First: 578 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 13,670 kg/m3 W/mK
Conductivity, Thermal: 10 W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1944
95
2
8
18
32
25
8
2
Am
Americium
(243)
Element: Curium
Series: Actinoids
Atomic Number: 96
Energy Levels: 2, 8, 18, 32, 25, 9, 2
Atomic Weight: (247) u
Electronegativity: 1.3
Melting Point: 1,345° C
Boiling Point: 3,110° C
Electron Affinity: N/A kJ/mol
Ionization, First: 581 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 13,510 kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19442
Series: Actinoids
Atomic Number: 96
Energy Levels: 2, 8, 18, 32, 25, 9, 2
Atomic Weight: (247) u
Electronegativity: 1.3
Melting Point: 1,345° C
Boiling Point: 3,110° C
Electron Affinity: N/A kJ/mol
Ionization, First: 581 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 13,510 kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1944
96
2
8
18
32
25
9
2
Cm
Curium
(247)
Element: Berkelium
Series: Actinoids
Atomic Number: 97
Energy Levels: 2, 8, 18, 32, 27, 8, 2
Atomic Weight: (247) u
Electronegativity: 1.3
Melting Point: 1,050° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 601 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 14,780 kg/m3 W/mK
Conductivity, Thermal: 10 W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19492
Series: Actinoids
Atomic Number: 97
Energy Levels: 2, 8, 18, 32, 27, 8, 2
Atomic Weight: (247) u
Electronegativity: 1.3
Melting Point: 1,050° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 601 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 14,780 kg/m3 W/mK
Conductivity, Thermal: 10 W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1949
97
2
8
18
32
27
8
2
Bk
Berkelium
(247)
Element: Californium
Series: Actinoids
Atomic Number: 98
Energy Levels: 2, 8, 18, 32, 28, 8, 2
Atomic Weight: (251) u
Electronegativity: 1.3
Melting Point: 899.9° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 608 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 15,100 kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19502
Series: Actinoids
Atomic Number: 98
Energy Levels: 2, 8, 18, 32, 28, 8, 2
Atomic Weight: (251) u
Electronegativity: 1.3
Melting Point: 899.9° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 608 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: 15,100 kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1950
98
2
8
18
32
28
8
2
Cf
Californium
(251)
Element: Einsteinium
Series: Actinoids
Atomic Number: 99
Energy Levels: 2, 8, 18, 32, 29, 8, 2
Atomic Weight: (252) u
Electronegativity: 1.3
Melting Point: 859.9° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 619 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19522
Series: Actinoids
Atomic Number: 99
Energy Levels: 2, 8, 18, 32, 29, 8, 2
Atomic Weight: (252) u
Electronegativity: 1.3
Melting Point: 859.9° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 619 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1952
99
2
8
18
32
29
8
2
Es
Einsteinium
(252)
Element: Fermium
Series: Actinoids
Atomic Number: 100
Energy Levels: 2, 8, 18, 30, 28, 8, 2
Atomic Weight: (257) u
Electronegativity: 1.3
Melting Point: 1,500° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 627 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19522
Series: Actinoids
Atomic Number: 100
Energy Levels: 2, 8, 18, 30, 28, 8, 2
Atomic Weight: (257) u
Electronegativity: 1.3
Melting Point: 1,500° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 627 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1952
100
2
8
18
30
28
8
2
Fm
Fermium
(257)
Element: Mendelevium
Series: Actinoids
Atomic Number: 101
Energy Levels: 2, 8, 18, 32, 31, 8, 2
Atomic Weight: (258) u
Electronegativity: 1.3
Melting Point: 830° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 635 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19552
Series: Actinoids
Atomic Number: 101
Energy Levels: 2, 8, 18, 32, 31, 8, 2
Atomic Weight: (258) u
Electronegativity: 1.3
Melting Point: 830° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 635 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1955
101
2
8
18
32
31
8
2
Md
Mendelevium
(258)
Element: Nobelium
Series: Actinoids
Atomic Number: 102
Energy Levels: 2, 8, 18, 32, 32, 8, 2
Atomic Weight: (259) u
Electronegativity: 1.3
Melting Point: 830° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 642 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19582
Series: Actinoids
Atomic Number: 102
Energy Levels: 2, 8, 18, 32, 32, 8, 2
Atomic Weight: (259) u
Electronegativity: 1.3
Melting Point: 830° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 642 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1958
102
2
8
18
32
32
8
2
No
Nobelium
(259)
Element: Lawrencium
Series: Actinoids
Atomic Number: 103
Energy Levels: 2, 8, 18, 32, 32, 8, 3
Atomic Weight: (266) u
Electronegativity: 1.3
Melting Point: 1,600° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 470 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 19612
Series: Actinoids
Atomic Number: 103
Energy Levels: 2, 8, 18, 32, 32, 8, 3
Atomic Weight: (266) u
Electronegativity: 1.3
Melting Point: 1,600° C
Boiling Point: N/A° C
Electron Affinity: N/A kJ/mol
Ionization, First: 470 kJ/mol
Radius Calculated: N/A pm
Hardness, Brinell: N/A MPa
Modulus, Bulk: N/A GPa
Density, STP: N/A kg/m3 W/mK
Conductivity, Thermal: N/A W/mK
Heat, Specific: N/A J/kgK
Abundance, Universe: 0 %
Discovered: 1961
103
2
8
18
32
32
8
3
Lr
Lawrencium
(266)
Notes
*For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses.
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers (mass numbers) due to different numbers of neutrons in their nuclei. While all isotopes of a given element have almost the same chemical properties, they have different atomic masses and physical properties.
Compounds A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) composed of atoms from more than one element held together by chemical bonds. A molecule consisting of atoms of only one element is therefore not a compound.
There are four types of compounds, depending on how the constituent atoms are held together:
A chemical formula specifies the number of atoms of each element in a compound molecule, using the standard abbreviations for the chemical elements and numerical subscripts. For example, a water molecule has formula H2O indicating two hydrogen atoms bonded to one oxygen atom. Many chemical compounds have a unique CAS number identifier assigned by the Chemical Abstracts Service. Globally, more than 350,000 chemical compounds (including mixtures of chemicals) have been registered for production and use.
A compound can be converted to a different chemical substance by interaction with a second substance via a chemical reaction. In this process, bonds between atoms may be broken in either or both of the interacting substances, and new bonds formed.
Electron Shells In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus. The closest shell to the nucleus is called the "1 shell" (also called the "K shell"), followed by the "2 shell" (or "L shell"), then the "3 shell" (or "M shell"), and so on farther and farther from the nucleus. The shells correspond to the principal quantum numbers (n = 1, 2, 3, 4 ...) or are labeled alphabetically with the letters used in X-ray notation (K, L, M, …).
Each shell can contain only a fixed number of electrons: The first shell can hold up to two electrons, the second shell can hold up to eight (2 + 6) electrons, the third shell can hold up to 18 (2 + 6 + 10) and so on. The general formula is that the nth shell can in principle hold up to 2(n2) electrons.[1] For an explanation of why electrons exist in these shells see electron configuration.
Each shell consists of one or more subshells, and each subshell consists of one or more atomic orbitals.
Electron Configuration In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals.[1] For example, the electron configuration of the neon atom is 1s2 2s2 2p6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively.
Electronic configurations describe each electron as moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.
According to the laws of quantum mechanics, for systems with only one electron, a level of energy is associated with each electron configuration and in certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.
Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. This is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.
Pnictogen - A pnictogen[1] (/ˈpnɪktədʒən/ or /ˈnɪktədʒən/; from Ancient Greek: πνῑ́γω "to choke" and -gen, "generator") is any of the chemical elements in group 15 of the periodic table. This group is also known as the nitrogen family. It consists of the elements nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb), bismuth (Bi), and perhaps the chemically uncharacterized synthetic element moscovium (Mc).
In modern IUPAC notation, it is called Group 15. In CAS and the old IUPAC systems it was called Group VA and Group VB respectively (pronounced "group five A" and "group five B", "V" for the Roman numeral 5).[2] In the field of semiconductor physics, it is still usually called Group V.[3] The "five" ("V") in the historical names comes from the "pentavalency" of nitrogen, reflected by the stoichiometry of compounds such as N2O5. They have also been called the pentels.
Chalcogen - The chalcogens (ore forming) (/ˈkælkədʒənz/ KAL-kə-jənz) are the chemical elements in group 16 of the periodic table. This group is also known as the oxygen family. It consists of the elements oxygen (O), sulfur (S), selenium (Se), tellurium (Te), and the radioactive element polonium (Po). The chemically uncharacterized synthetic element livermorium (Lv) is predicted to be a chalcogen as well.[1] Often, oxygen is treated separately from the other chalcogens, sometimes even excluded from the scope of the term "chalcogen" altogether, due to its very different chemical behavior from sulfur, selenium, tellurium, and polonium. The word "chalcogen" is derived from a combination of the Greek word khalkόs (χαλκός) principally meaning copper (the term was also used for bronze/brass, any metal in the poetic sense, ore or coin),[2] and the Latinized Greek word genēs, meaning born or produced.
Sulfur has been known since antiquity, and oxygen was recognized as an element in the 18th century. Selenium, tellurium and polonium were discovered in the 19th century, and livermorium in 2000. All of the chalcogens have six valence electrons, leaving them two electrons short of a full outer shell. Their most common oxidation states are −2, +2, +4, and +6. They have relatively low atomic radii, especially the lighter ones.
Lighter chalcogens are typically nontoxic in their elemental form, and are often critical to life, while the heavier chalcogens are typically toxic.[1] All of the naturally occurring chalcogens have some role in biological functions, either as a nutrient or a toxin. Selenium is an important nutrient (among others as a building block of selenocysteine) but is also commonly toxic.[6] Tellurium often has unpleasant effects (although some organisms can use it), and polonium (especially the isotope polonium-210) is always harmful as a result of its radioactivity.
Sulfur has more than 20 allotropes, oxygen has nine, selenium has at least eight, polonium has two, and only one crystal structure of tellurium has so far been discovered. There are numerous organic chalcogen compounds. Not counting oxygen, organic sulfur compounds are generally the most common, followed by organic selenium compounds and organic tellurium compounds. This trend also occurs with chalcogen pnictides and compounds containing chalcogens and carbon group elements.
Oxygen is generally obtained by separation of air into nitrogen and oxygen. Sulfur is extracted from oil and natural gas. Selenium and tellurium are produced as byproducts of copper refining. Polonium and livermorium are most available in particle accelerators. The primary use of elemental oxygen is in steelmaking. Sulfur is mostly converted into sulfuric acid, which is heavily used in the chemical industry.[6] Selenium's most common application is glassmaking. Tellurium compounds are mostly used in optical disks, electronic devices, and solar cells. Some of polonium's applications are due to its radioactivity.[
Halogen - The halogens (/ˈhælədʒən, ˈheɪ-, -loʊ-, -ˌdʒɛn/[1][2][3]) are a group in the periodic table consisting of five chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). The artificially created element 117, tennessine (Ts), may also be a halogen. In the modern IUPAC nomenclature, this group is known as group 17.
The name "halogen" means "salt-producing". When halogens react with metals, they produce a wide range of salts, including calcium fluoride, sodium chloride (common table salt), silver bromide and potassium iodide.
The group of halogens is the only periodic table group that contains elements in three of the main states of matter at standard temperature and pressure. All of the halogens form acids when bonded to hydrogen. Most halogens are typically produced from minerals or salts. The middle halogens—chlorine, bromine, and iodine—are often used as disinfectants. Organobromides are the most important class of flame retardants, while elemental halogens are dangerous and can be lethally toxic.
State of Matter
In physics, a state of matter is one of the distinct forms in which matter can exist. Four states of matter are observable in everyday life: solid, liquid, gas, and plasma. Many intermediate states are known to exist, such as liquid crystal, and some states only exist under extreme conditions, such as Bose–Einstein condensates, neutron-degenerate matter, and quark–gluon plasma, which only occur, respectively, in situations of extreme cold, extreme density, and extremely high energy. For a complete list of all exotic states of matter, see the list of states of matter.
Historically, the distinction is made based on qualitative differences in properties. Matter in the solid state maintains a fixed volume and shape, with component particles (atoms, molecules or ions) close together and fixed into place. Matter in the liquid state maintains a fixed volume, but has a variable shape that adapts to fit its container. Its particles are still close together but move freely. Matter in the gaseous state has both variable volume and shape, adapting both to fit its container. Its particles are neither close together nor fixed in place. Matter in the plasma state has variable volume and shape, and contains neutral atoms as well as a significant number of ions and electrons, both of which can move around freely.
The term phase is sometimes used as a synonym for state of matter, but a system can contain several immiscible phases of the same state of matter.
Four Fundamental States
Solid - In a solid, constituent particles (ions, atoms, or molecules) are closely packed together. The forces between particles are so strong that the particles cannot move freely but can only vibrate. As a result, a solid has a stable, definite shape, and a definite volume. Solids can only change their shape by an outside force, as when broken or cut.
In crystalline solids, the particles (atoms, molecules, or ions) are packed in a regularly ordered, repeating pattern. There are various different crystal structures, and the same substance can have more than one structure (or solid phase). For example, iron has a body-centred cubic structure at temperatures below 912 °C (1,674 °F), and a face-centred cubic structure between 912 and 1,394 °C (2,541 °F). Ice has fifteen known crystal structures, or fifteen solid phases, which exist at various temperatures and pressures.
Glasses and other non-crystalline, amorphous solids without long-range order are not thermal equilibrium ground states; therefore they are described below as nonclassical states of matter.
Solids can be transformed into liquids by melting, and liquids can be transformed into solids by freezing. Solids can also change directly into gases through the process of sublimation, and gases can likewise change directly into solids through deposition.
Liquid - A liquid is a nearly incompressible fluid that conforms to the shape of its container but retains a (nearly) constant volume independent of pressure. The volume is definite if the temperature and pressure are constant. When a solid is heated above its melting point, it becomes liquid, given that the pressure is higher than the triple point of the substance. Intermolecular (or interatomic or interionic) forces are still important, but the molecules have enough energy to move relative to each other and the structure is mobile. This means that the shape of a liquid is not definite but is determined by its container. The volume is usually greater than that of the corresponding solid, the best known exception being water, H2O. The highest temperature at which a given liquid can exist is its critical temperature.
Gas - A gas is a compressible fluid. Not only will a gas conform to the shape of its container but it will also expand to fill the container.
In a gas, the molecules have enough kinetic energy so that the effect of intermolecular forces is small (or zero for an ideal gas), and the typical distance between neighboring molecules is much greater than the molecular size. A gas has no definite shape or volume, but occupies the entire container in which it is confined. A liquid may be converted to a gas by heating at constant pressure to the boiling point, or else by reducing the pressure at constant temperature.
At temperatures below its critical temperature, a gas is also called a vapor, and can be liquefied by compression alone without cooling. A vapor can exist in equilibrium with a liquid (or solid), in which case the gas pressure equals the vapor pressure of the liquid (or solid).
A supercritical fluid (SCF) is a gas whose temperature and pressure are above the critical temperature and critical pressure respectively. In this state, the distinction between liquid and gas disappears. A supercritical fluid has the physical properties of a gas, but its high density confers solvent properties in some cases, which leads to useful applications. For example, supercritical carbon dioxide is used to extract caffeine in the manufacture of decaffeinated coffee.
Plasma - Like a gas, plasma does not have definite shape or volume. Unlike gases, plasmas are electrically conductive, produce magnetic fields and electric currents, and respond strongly to electromagnetic forces. Positively charged nuclei swim in a "sea" of freely-moving disassociated electrons, similar to the way such charges exist in conductive metal, where this electron "sea" allows matter in the plasma state to conduct electricity.
A gas is usually converted to a plasma in one of two ways, e.g., either from a huge voltage difference between two points, or by exposing it to extremely high temperatures. Heating matter to high temperatures causes electrons to leave the atoms, resulting in the presence of free electrons. This creates a so-called partially ionised plasma. At very high temperatures, such as those present in stars, it is assumed that essentially all electrons are "free", and that a very high-energy plasma is essentially bare nuclei swimming in a sea of electrons. This forms the so-called fully ionised plasma.
The plasma state is often misunderstood, and although not freely existing under normal conditions on Earth, it is quite commonly generated by either lightning, electric sparks, fluorescent lights, neon lights or in plasma televisions. The Sun's corona, some types of flame, and stars are all examples of illuminated matter in the plasma state.
Phase Transitions
A state of matter is also characterized by phase transitions. A phase transition indicates a change in structure and can be recognized by an abrupt change in properties. A distinct state of matter can be defined as any set of states distinguished from any other set of states by a phase transition. Water can be said to have several distinct solid states. The appearance of superconductivity is associated with a phase transition, so there are superconductive states. Likewise, ferromagnetic states are demarcated by phase transitions and have distinctive properties. When the change of state occurs in stages the intermediate steps are called mesophases. Such phases have been exploited by the introduction of liquid crystal technology.
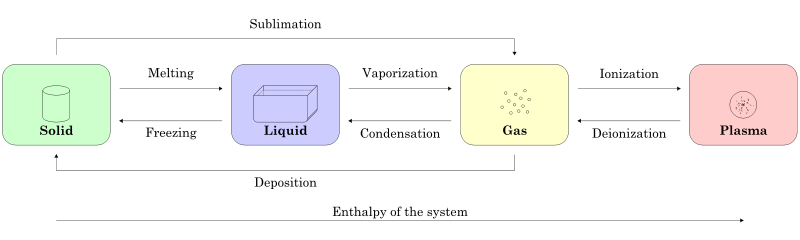
The state or phase of a given set of matter can change depending on pressure and temperature conditions, transitioning to other phases as these conditions change to favor their existence; for example, solid transitions to liquid with an increase in temperature. Near absolute zero, a substance exists as a solid. As heat is added to this substance it melts into a liquid at its melting point, boils into a gas at its boiling point, and if heated high enough would enter a plasma state in which the electrons are so energized that they leave their parent atoms.
Forms of matter that are not composed of molecules and are organized by different forces can also be considered different states of matter. Superfluids (like Fermionic condensate) and the quark–gluon plasma are examples.
In a chemical equation, the state of matter of the chemicals may be shown as (s) for solid, (l) for liquid, and (g) for gas. An aqueous solution is denoted (aq). Matter in the plasma state is seldom used (if at all) in chemical equations, so there is no standard symbol to denote it. In the rare equations that plasma is used it is symbolized as (p).
Isotopes
An isotope is an atom of an element with the same number of protons but a different number of neutrons. This gives an equal proton number (so by definition it's the same element) but a different mass number. The discovery of isotopes refuted Dalton's claim of atoms that all atoms of an element were identical – this is not true!
Isotopes are why the periodic table contains decimals for many elements' relative masses. Relative atomic mass for an element is an average value account for the masses and relative abundance of each isotope of an element.
"Relative" when talking about the mass of any atom or molecule, means relative to an atom of carbon-12.
Because neutrons have no charge, the number of neutrons doesn't change an atom's chemical reactivity. Therefore isotopes of an element have identical chemical properties to each other isotope!
Because neutrons have a relative atomic mass of 1 amu (the same as protons), isotopes do affect the relative atomic mass of elements as they are written in the periodic table. Ice cubes made of normal water (H2O) are less dense than liquid water. Ice cubes made with deuterated water (D2O), where the hydrogen atoms are hydrogen-2 atoms, sink in regular liquid water!
Chemical Compounds
A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) composed of atoms from more than one element held together by chemical bonds. A molecule consisting of atoms of only one element is therefore not a compound.
There are four types of compounds, depending on how the constituent atoms are held together:
- Molecules held together by covalent bonds
- Ionic compounds held together by ionic bonds
- Intermetallic compounds held together by metallic bonds
- Certain complexes held together by coordinate covalent bonds







